Page 1 :
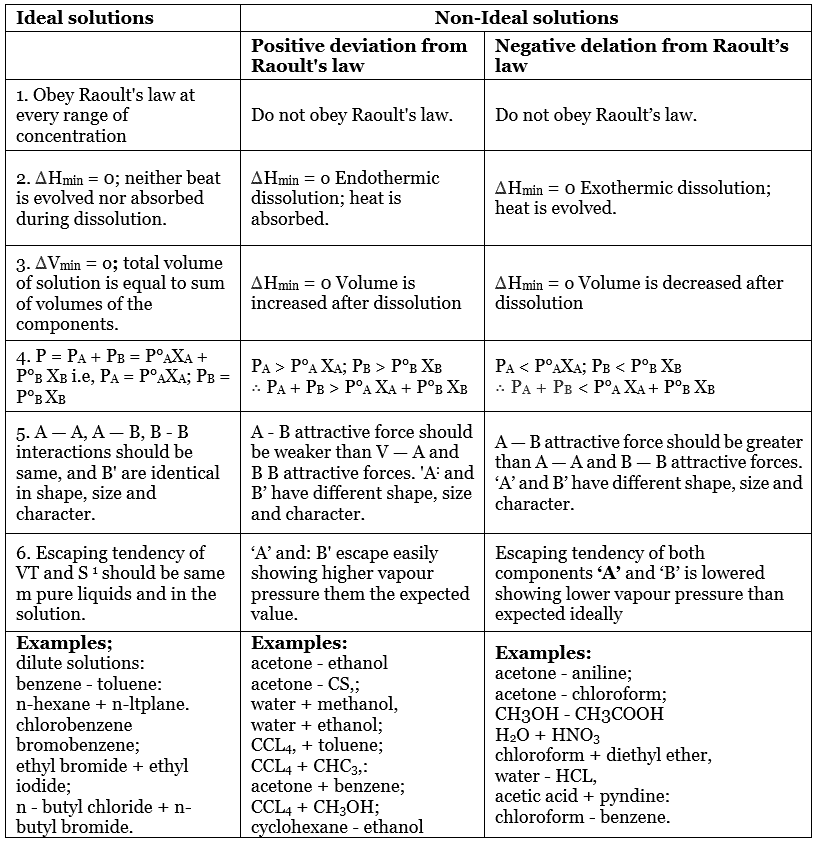
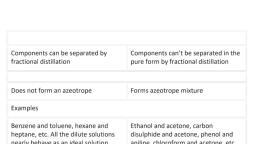
Ideal solutions, , Non-Ideal solutions, , , , Positive deviation from, Raoult's law, , Negative delation from Raoult’s, law, , , , 1. Obey Raoult’s law at, every range of, concentration, , Do not obey Raoult's law., , Do not obey Raoult’s law., , , , 2. AHmin = 0; neither beat, is evolved nor absorbed, during dissolution., , AHnmin = 0 Endothermic, dissolution; heat is, absorbed., , AHmin = 0 Exothermic dissolution;, heat is evolved., , , , 3. AVinin = 0; total volume, of solution is equal to sum, of volumes of the, components., , AHnin = 0 Volume is, increased after dissolution, , AHnin = 0 Volume is decreased after, dissolution, , , , 4.P = Pa+ Pp= P°aXa+, P°s Xpie, Pa = P°aXa; Pp=, Pop Xe, , , , Pa > P°s Xa; Pp > P°s XB, - Pa+ Pp> Pa Xa+ Pop Xp, , Pa < P°aXa; Pp < P°s XB, - Pa + Pp < P°s Xa+ P's Xp, , , , 5.A—A,A—B,B-B, interactions should be, same, and B' are identical, in shape, size and, character., , A - B attractive force should, be weaker than V— Aand, B B attractive forces. 'A‘ and, B’ have different shape, size, and character., , A~—Battractive force should be greater, than A— Aand B — B attractive forces., ‘’ and B’ have different shape, size and, character., , , , 6. Escaping tendency of, VT and $ * should be same, m pure liquids and in the, solution., , ‘a’ and: B' escape easily, showing higher vapour, pressure them the expected, value., , Escaping tendency of both, components ‘A’ and ‘B’ is lowered, showing lower vapour pressure than, expected ideally, , , , , , Examples;, dilute solutions:, benzene - toluen:, n-hexane + n-ltplane., chlorobenzene, bromobenzene;, , ethyl bromide + ethyl, iodide;, , n-butyl chloride + nbutyl bromide., , , , , , Examples:, , acetone - ethanol, acetone - CS,;, , water + methanol,, water + ethanol;, CCLy, + toluene;, CCL; + CHC;,:, acetone + benzene;, CCL, + CH;0H;, cyclohexane - ethanol, , , , Examples:, , acetone - aniline;, , acetone - chloroform;, CH30H - CH3COOH, , H.0 + HNO;, , chloroform + diethyl ether,, water - HCL,, , acetic acid + pyndine:, chloroform - benzene.
 Learn better on this topic
Learn better on this topic






































 Learn better on this topic
Learn better on this topic









