Page 1 :
Unit 13-NITROGEN CONTAINING ORGANIC COMPOUNDS, Two marks:, 1. Name the product obtained when a nitrile is reduced by H2/Ni, . Give the equation., H2 / Ni,, Ans. Primary amine: RCN , RCH2NH2., 2. How is nitrobenzene converted into aniline. Give the equation., Ans. By reduction using Sn/ HCl, , 3. What are A and B?, , Ans. A is RCH2NH2, B is RNH2, 4. Give the equation which will be an example for Hofmann bromamide reaction., Ans. CH3CONH2 + Br2 + 4NaOH , CH3NH2 + 2NaBr + Na2CO3 + 2H2O, Acetamide, methanamine, 5. Gabriel phthalimide synthesis is used to prepare which class of organic compound?, Aniline cannot be prepared by this method. Give reason., Ans. 1° aliphatic amine, Aryl halides are not reactive towards nucleophilic substitution reaction., 6. Name the reaction by which a 1° amine is prepared from an amide having one carbon, atom more than 1° amine. Give the general equation., Ans. Hofmann bromamide degradation reaction, RCONH2 + Br2 + 4KOH , 2NH2 + 2KBr + K2CO3 + 2H2O, 7. Between CH3CH2CH2NH2 and (CH3)3N, which has higher boiling point and why?, Ans. CH3CH2CH2NH2 has higher boiling point. CH3CH2CH2NH2 has more H atoms on N, to form intermolecular hydrogen bonding., 8. Give reason: i) Amines have lower boiling point than alcohol of same molar mass., ii) Amines are insoluble in water., Ans. i) Nitrogen in amines is less electronegative than oxygen in alcohol. Hence amines do, not form H-bonds among them., ii) Amines do not form H bonds with water., 9. Amines are both Bronsted base and Lewis base. How?, Ans. Amines can accept H+, hence Bronsted bases. Amines can donate a pair of electrons, hence Lewis bases., , 1
Page 3 :
20. Identify the main organic product in the following reactions:, NaNO2 /HCl, i) C6H5NH2 + HNO2 , ________, 0 C, Br2 / NaOH, ii) C6H5CONH2 , __________, , Ans. i) C6H5N2Cl, , ii) C6H5NH2, , 21. What is benzene sulphonyl chloride also known as? An amine with benzene sulphonyl, chloride forms a compound insoluble in an alkali. Identify the class of the amine., Ans. Hinsberg’s reagent. 2° amine., 22. How does Hinsberg’s reagent help to distinguish 1° amine and a 2° amine? Explain., Ans. The given amine is treated with Hinsberg’s reagent. If the product formed is soluble in, an alkali, the amine is 1°. If the product formed is insoluble in an alkali, the amine is 2°., 23. Complete the following equations:, , Ans., , 24. What is the significance of acetylation of aniline before nitrating it?, Ans. When aniline is treated with concentrated HNO3, much of the aniline gets oxidized,, aniline gets protonated and the major product is meta-nitroaniline. Hence to avoid all, this aniline is acetylated. Acetylated aniline, avoids oxidation of aniline and controlled, nitration yields p-nitro aniline as the major product., 25. Give reasons: i) aniline does not undergo Friedel-Crafts reaction. ii) aniline with, concentrated HNO3 forms meta nitro compound in significant amounts., Ans. i) Aniline reacts with AlCl3 to form a salt, which makes nitrogen of aniline to get a, positive charge, which becomes a strongly deactivating group., ii) Aniline with conc. HNO3 forms anilinium ion which is meta directing., 26. What is diazotization? Give the general formula of a diazonium salt., Ans. Conversion of 1° aromatic amine into diazonium salt is diazotization., , , , , General formula: Ar N 2 X, , , , or, , , , R N 2 X , where R = Ar, , 3
Page 4 :
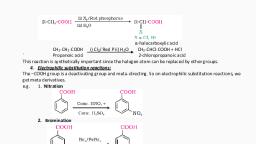
27. How is benzene diazonium chloride prepared from aniline? Give the equation., Ans. It is prepared by the reaction of aniline with nitrous acid (NaNO2/ HCl) at 0°C, , , , , 0 C, C6H5NH2 + NaNO2 + 2HCl , C6 H5 N 2 Cl + NaCl + 2H2O, , 28. What is Sandmeyer’s reaction? Give an example., Ans. Replacement of diazonium group by Cl/ Br in presence of Cu(I) ion., , , , , Cu 2Cl2 /HCl, E.g.: Ar N 2 X , ArCl + N2, , 29. Name the organic products obtained in the following reactions:, , , , , CuCN/KCN, i) Ar N 2 X , __________ + N2, , , , , ii) Ar N 2 Cl + H3PO2 + H2O , _____ + N2 + CH3CHO + HCl, Ans. i) Aryl cyanide, , ii) Benzene, , 30. How is a diazonium salt converted into iodobenzene? Give the equation., Ans. By treating diazonium salt with potassium iodide., , , , , ArI + KCl + N2, Ar N 2 X + KI , , iodobenzene, 31. Give an example for a coupling reaction with an equation., Ans. Benzene diazonium chloride reacts with phenol to form p-hydroxyazobenze. This is an, example for coupling reaction., , 32. How is benzene diazonium chloride converted into an azo dye? Give an example for an, azo dye., Ans. Azo dyes are the products obtained when reaction of benzene diazonium chloride with, phenol or aniline takes place with retention of diazo group. E.g.: benzene diazonium, chloride couples with aniline to form an azo dye p-amino azo benzene (yellow dye), 33. Mention the importance of diazonium salt in synthetic organic chemistry., Ans. i) Aryl fluoride and iodides that cannot be prepared by direct halogenation can be, synthesized., ii) It helps to introduce many functional groups into aromatic ring, which cannot be, done by direct methods., THREE MARKS:, 34. Identify the X, Y, Z in the following:, , Ans. X is aniline, Y is benzene diazonium chloride, Z is iodobenzene, , 4
Page 5 :
35. Give equations for the preparation of methylamine (methanamine) by Gabrielphthalimide synthesis., Ans., , CHCl /Alc.KOH, H / Ni,, 36. RCN , Y. Y is a three carbon compound. What is R in RCN, X, X , and Y?, Ans. R = CH3, X = CH3CH2NH2, Y = CH3CH2NC, 2, , 3, , 37. Give equation for the conversion of aniline into 4-bromoaniline., Ans., , 38. An organic compound with formula C2H7N does not answer carbylamine test, but give a, product that is insoluble in an alkali, with Hinsberg reagent. Give the IUPAC name of X, and to what class of organic compound does it belong to?, Ans. X is CH3NHCH3., IUPAC name : N-methylmethanamine., It is a 2° amine., NaNO /HCl, warm, 39. X , Y , Z. Y + Z , orange dye (p-hydroxyazobenzene). What are X,, 0 C, 2, , Y and Z?, Ans., , 5