Page 1 :
CHAPTER AT A GLANCE, , ‘Pure’ word means that there is no mixing in a substance. But, Is Matter Around Us Pure ?, , 9
Page 2 :
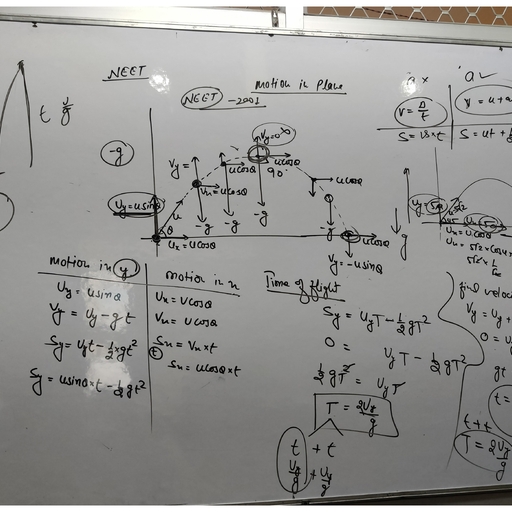
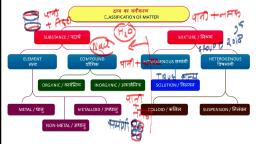
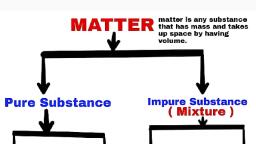
according to scientific language all things are mixture of so, many substances, not of single one. That’s why they are not pure., E.g., Milk, water, fat, etc., • Pure substances means that all elements have same chemical properties., • A pure substance is made up of same kind of elements., Substance : A substance is a kind of matter that cannot be separated into other, kind of matter by any physical process. A pure substance is made up of same, kind of elements., What is a mixture ?, It is a substance in which two or more substances (element or compound) are, simply mixed together in any proportion. Examples : The air is a mixture of, oxygen, nitrogen, argon, carbon dioxide and water vapour., Types of Mixture : Mixture is of two types :, (i), Homogenous mixture, (ii), Heterogenous mixture, Homogenous Mixture : It has no visible boundaries of separation between the, various constituents, so they are called homogenous mixtures., Example : Sugar in water. It has a uniform composition throughout its, mass., Heterogenous Mixture : These types of mixtures has visible boundaries of, separation between the various constituents., Example : Mixture of sugar and sand. It does not have a uniform, composition throughout its mass., Solution : A solution is a homogenous mixture of two or more substances. E.g.,, Nimboo pani, soda water., Solution : A solution has a solvent and a solute as its components. The component, of the solution that dissolves the other component in it is called the solvent. The, component of the solution that is dissolved in the solvent is called the solute., , Solution, Solute Solvent, (Substance dissolved) , (Substance in which dissolved), E.g.,, , 10, , Sugar , , +, , Science Class (SA-I)- IX, , Water , , ⇒, , Sugar solution
Page 3 :
Types of Solution, True, 1. Size of solute, particles smallest., < 10-9 m., , Colloidal, 1. Size of solute particles bigger than, true but smaller than suspension. In, between 10-9 to 10-6 m., , Suspension, 1. Size of particles, biggest. > 10-6 m., , 2. Solute particles, can’t be seen with, naked eye., 3. Homogenous, mixture., , 2. Solute particles can’t be seen with, Naked eye., , 2. Can be seen, with naked eye., , 3. Seems homogenous but actually, heterogenous mixture., , 3. Heterogenous, mixture., , 4. Particles can’t, be separated by, filteration., , 4. Particles can’t be separated by, filteration., , 4. Can be, separated by, filteration., , 5. Transparent, , 5. Translucent, , 5. Opaque, , 6. Stable solutions 6. Stable solutions., – i.e., solute, particles do not, settle on keeping., , 6.Unstable, solution – solute, particles settle, upon keeping., , 7. Do not show, tyndall effect., , 7. Show tyndall effect., , 8. Solution diffuse, rapidly through, filter paper as, well as parchment, paper., 9. e.g., Sugar in, water., , 8. Colloid particles pass through, filter paper but not through, parchment paper., , 7. May or may, not show tyndall, effect., 8. Suspension, particles do not, pass through filter, paper as well as, parchment paper., 9. e.g., Sand/mud, in water., , 9. e.g., Milk, blood., Common examples of colloids :, , Dispersal Phase Dispersion, Type, Example, (Solute) Medium, (Solvent), 1. Liquid , Gas Aerosol Fog, cloud, 2. Solid , Gas Aerosol, Smoke, Is Matter Around Us Pure ?, , 11
Page 4 :
3. Gas , Liquid , Foam, Shaving , , cream, 4. Liquid, Liquid Emulsion, Milk, face, , cream, , emulsion paint, 5. Solid, Liquid , Sol Mud, digene, 6. Gas , Solid , Foam Foam, rubber,, , sponge, 7. Liquid, Solid , Gel Jelly, cheese, 8. Solid, Solid Solid sol, Coloured , , gemstones,, , glass (milky,, coloured), • Gas in gas is not a colloidal solution – it is called a mixture., Concentration of Solution, 1., , Mass by mass percentage , , =, , 2., , Mass by volume percentage , , =, , Methods of Separation of Mixtures, , (1), , Evaporation :, , (2), , Centrifugation :, , Basic principle : Out of the two components of a mixture one can, evaporate [i.e., has less boiling point] and other has higher boiling point., Example : Mixture of dye [higher boiling point] and water. Out of water, , and dye, water evaporates but dye is left behind in petri dish., , 12, , Basic principle : When mixture is rotated very fast, then denser particles, Science Class (SA-I)- IX
Page 5 :
are forced at the bottom and lighter particles stay above., , Example : Separating cream from milk., Can you think what is toned and double toned milk ?, Applications :, (a), Used in diagnostic labs for blood and urine tests., (b), Used in dairies and home to separate butter from cream., (c), Used in washing machines dryers to squeeze out water from, , clothes., Have you seen dust particles from our clothes settle at the bottom of, washing tub. Do you know why now ?, , (3), , Differential entraction :, , Basic principle : Two immiscible liquids (which do not dissolve in, each other) can be easily separated by putting in a differential entraction funnel/, separating funnel., Example : Water from oil can be separated by first opening the stop cock, till water is removed in one beaker, then afterwards oil can be collected in a, separate beaker., , Is Matter Around Us Pure ?, , 13
Page 6 :
Applications :, (a), Separation of oil from water., (b), Extraction of iron from its ore. Lighter slag is removed from, , above the molten iron., , (4), , Sublimation :, , Basic principle : Out of the two components, one will sublime (directly, converts to gas from solid) and other will not., Example : NH4Cl (ammonium chloride) and NaCl common salt mixture can, be easily separated by heating so that NH4Cl sublimes but common salt, remains behind., , Applications :, (a), Camphor, naphthalene, anthracene, NH4Cl can sublime., , (5), , Chromatography :, , Basic principle : Coloured components of a mixture can be separated by, using an adsorbent on which they are adsorbed at different rates., (Absorption is the process of surface absorption.), , When water/any suitable solvent moves up, the chromatography paper, ink with two different colours separates because both colours are adsorbed at, different speeds., 14, , Science Class (SA-I)- IX
Page 7 :
Applications :, (a), To separate colours of a dye., (b), To separate pigments from natural colours like chlorophyll., (c), To separate drugs from blood., (Can you guess what is done when athletes undergo a doping test for, their blood ?), , (6), , Distillation :, , Basic principle : Out of the two components one has a lower boiling, point and other has higher boiling point. This is used to separate two or more, miscible liquids., , Example : When mixture of acetone and water is heated, acetone having, lesser boiling point, boils and moves to delivery tube, within which it condenses, back to liquid with the help of a condenser clamped to it. Thus, acetone is, separated out in a beaker and water is left in the distillation flask., Note : If there are more than two components (liquids) mixed (with, different boiling points) then we use a fractionating column to separate all the, components from each other. This process is done for air, petroleum etc. , , Is Matter Around Us Pure ?, , 15
Page 8 :
Petroleum is separated into, paraffin wax, lubricating oil,, diesel, kerosene, petrol and, petrol gas by this method., Fractional Distillation of Air :, Air is also separated by this method., , Some of the applications of fractional distillation :, (a), In petroleum refineries, petrochemical and chemical plants,, , natural gas processing and cryogenic air separation plants., (b), In oil refineries to separate crude oil into useful substances (or, , fractions)., (c), In the process of organic juice., (d), In the separation of oxygen, liquid nitrogen and argon from air., , (7), , Crystallisation :, , Basic principle : To remove impurities from a mixture by first dissolving, in a suitable solvent and then crystallising out one component., For example : Copper sulphate crystals (impure) are first dissolved in, sulphuric acid and then heated to saturated solution. Now, this solution, is left overnight. So, only pure copper sulphate crystals are formed, whereas impurities are left behind in the solution. This solution can be, thus filtered so as to get pure copper sulphate crystals on filter paper., Why is crystallisation better than evaporation ?, (i), Some solids decompose or get charred upon heating to dryness, , during evaporation. E.g., sugar., (ii), Some impurities remain dissolved in solution after filteration. On, , evaporation, these impurities do not evaporate and remain with, the mixture., Applications :, (a), Purification of salt from sea water., (b), Separation of crystals [e.g., alum (phitkari, copper sulphate)], , from their impure crystals., 16, , Science Class (SA-I)- IX
Page 9 :
Water purification in water treatment plants, , Physical Vs Chemical Changes, Chemical, •, Not easily reversed, •, New product(s) formed, •, Reactants used up, •, Often heat/light/sound/fizzing, occurs, •, Electricity may be produced, •, A precipitate may form, •, E.g., Wood burning, , Physical, •, Easily reversible, •, No new products, •, Often just a state change, •, E.g., ice melting, , Elements, Made of same type of atoms, S. No. Metals, 1., Lustrous, , Non-metals, Non-lustrous, , Metalloids, Metallaoids have, intermediate properties, between metals and, non-metals., , Is Matter Around Us Pure ?, , 17
Page 10 :
2., , Malleable, ductile, , 3., 4., , Sonorous, Good conductors of, heat & electricity, E.g., Gold, iron etc., , 5., , Non-malleable, non- E.g., Boron, germanium,, ductile, silicon, Non-sonorous, Bad conductors, E.g., Oxygen,, phosphorus, , Mixture, 1. Elements or compounds are, simply mixed so no new, substance is formed., , Compound, 1. Substances are reacted together with, each other to make a new substance., , 2. Elements do not combine in a, fixed ratio., , 2. Composition of the components is, fixed i.e., they combine together in a, fixed ratio according to their masses., , 3. A mixture shows the properties 3. Compound doesn’t show the properties, of its components., of component elements., 4. Components can be easily, separated by any mechanical, method which is suitable., , 4. Components can’t be separated, from each other by simple mechanical, methods., , 5. E.g., Mixture of iron and, sulphur., , 5. E.g., Iron and sulphur react to form, iron sulphide., , Law of Constant Proportions, When a compound is formed two or more elements combine in a fixed ratio, according to their masses. For e.g., H2O has H : O = 1 : 8., Law of Conservation of Mass, Mass can neither be created no destroyed in a chemical reaction., , 18, , Science Class (SA-I)- IX
Page 11 :
QUESTIONS, VERY SHORT ANSWER TYPE QUESTIONS (1 Mark), 1., , Suggest methods used for separation ?, (a), , Husk from wheat, , (b), , Sand from water, , (c), , Stones from daal, , (d), , Camphor powder and common salt powder, , (e), , Butter from milk, , (f), , Pure potash alum from impure potash alum (phitkari), SHORT ANSWER TYPE QUESTIONS (2 Marks), , 1., , Write basic principles behind :, (a), , Chromatography, , (b), , Crystallisation, , (c), , Distillation, , (d), , Centrifugation, , 2., , Work out a process for separating a mixture of sand, NH4Cl and common, salt ?, , 3., , How can we separate alcohol from water ? Explain the whole process, with diagram ?, , 4., , The municipality water that we get at our homes may still have germs in, it. What can we do for it ?, , 5., , A student was having a beaker full of muddy water. He put a whitish, stone like substance in it and suddenly the mud settled down. What is this, white substance and what do we call this process ?, , Is Matter Around Us Pure ?, , 19
Page 12 :
SHORT ANSWER TYPE QUESTIONS (3 Marks), Explain the techniques used in following diagrams :, , 1., , 2., An athlete underwent a dope test and was found positive for drugs. What, could be the process used to identify that his blood contained those drugs ?, Explain the principle and process., , 20, , Science Class (SA-I)- IX
 Learn better on this topic
Learn better on this topic








































































 Learn better on this topic
Learn better on this topic









