Subjective Test of Class 10th, Science & Maths Periodic Classification Of Elements - Study Material
Page 1 :
Chapter, Practice, PART 1, Objective Questions, !, , Multiple Choice Questions, , 1. Dobereiner could identify how many triads from the, elements known at that time ?, (a) One, , (b) Two, , 9. Which of the following statements is not a correct, statement about the trends when going from left to, right across the periods of periodic table?, (NCERT), (a) The elements become less metallic in nature, (b) The number of valence electrons increases, (c) The atoms lose their electrons more easily, (d) The oxides become more acidic, , 10. Match the following columns :, , (c) Three, , (d) Four, , Column I, (Elements), , 2. Upto which element, the law of octaves was found to, be applicable?, (a) Oxygen, , (NCERT Exemplar), , (b) Calcium, , (c) Cobalt, , (d) Potassium, , 3. What type of oxide would Eka-aluminium form?, (NCERT Exemplar), , (a) EO3, , (b) E 3 O2, , (c) E 2 O3, , (d) EO, , 4. Which one of the following elements exhibit maximum, number of valence electrons?, (a) Na, , (b) Al, , (NCERT Exemplar), , (c) Si, , (d) P, , 5. Choose the correct statement., (a) Valency and valence electrons are always equal, (b) Valency is determined by using the number of valence, electrons, (c) Valence electrons are always equal to 8-valency, (d) Valency is always equal to 8-valence electrons, , 6. Elements with code letters Q and R occupy the, positions shown in the outline of the periodic table., , Column II, (Groups), , A. Be, , (i) Group 17, , B. F, , (ii) Group 15, , C. P, , (iii) Group 2, , D. Ar, , (iv) Group 18, , Codes, A B C D, (a) (iii) (i) (ii) (iv), (c) (ii) (iv) (iii) (i), , A B C D, (b) (i) (ii) (iii) (iv), (d) (iv) (ii) (i) (iii), , 11. Which metal is among the first ten elements in, modern periodic table?, (a) Lithium (Li), (c) Carbon (C), , (b) Boron (B), (d) Potassium (K), , 12. An element X is forming an acidic oxide. Its position, in modern periodic table will be, (a) group 1 and period 3, (c) group 13 and period 3, , (CBSE 2020), , (b) group 2 and period 3, (d) group 16 and period 3, , 13. An oxide of a metal shows no action with blue-litmus, but turns red-litmus to blue in its aqueous solution., The possible formula of the metal oxide can be, , R, Q, , (a) MgO, , What is the formula of the compound formed, between them?, (a) QR2, , (b) Q2R, , (c) Q2R3, , (d) Q3R2, , (b) CO2, , (c) SO2, , (d) H2O, , 14. The position of four elements A, B, C and D in the, modern periodic table are shown below. The, element which is most likely to form a basic oxide is, , 7. Which of the following statements is incorrect for, atomic size ?, (a) Atomic size of B > Be, (c) Atomic size of N > O, , (b) Atomic size of Be > B, (d) Atomic size of C > N, , B, , C, D, , 8. Which of the following gives the correct increasing, , A, , order of the atomic radii of O, F and N?, (NCERT Exemplar), , (a) O, F, N, , (b) N, F, O, , (c) O, N, F, , (d) F, O, N, , (a) B, , (b) C, , (c) A, , (d) D
Page 2 :
23, , CBSE Term II Science X, , h, , k, , Modern periodic law states that ‘‘the physical and, chemical properties of all the elements are the, periodic function of their atomic radius., The atomic radii of group 1 and group 2 elements, are given in the table., , i, , l, , Group 1 Atomic radii (nm) Group 2 Atomic radii (nm), , 15. Consider the part of periodic table given below:, Group/Period 1, I, , a, , II, , b, , III, , c, , IV, , d, , 2 13, , 14, , 15, , 16 17, , 18, j, , e, , g, f, , The most electropositive element is, (a) c, !, , (b) d, , (c) l, , (d) k, , Li, , 0.152, , Be, , 0.112, , Na, , 0.186, , Mg, , 0.160, , k, , 0.231, , Ca, , 0.197, , Assertion-Reasoning MCQs, , Rb, , 0.244, , Sr, , 0.215, , Direction (Q. Nos. 16-20) Each of these questions, contains two statements Assertion (A) and Reason (R)., Each of these questions also has four alternative choices,, any one of which is the correct answer. You have to select, one of the codes (a), (b), (c) and (d) given below., , Cs, , 0.262, , Ba, , 0.217, , Fr, , 0.270, , Ra, , 0.220, , (a) Both A and R are true and R is the correct explanation, of A., (b) Both A and R are true, but R is not the correct, explanation of A., (c) A is true, but R is false., (d) A is false, but R is true., , 16. Assertion Be, Mg and Ca can be classified as, , 17., , 18., 19., , 20., , Dobereiner’s triads., Reason Atomic mass of Mg is approximately the, average of the sum of atomic masses of Be and Ca., Assertion Sodium is an element of group 1 of the, modern periodic table., Reason All the elements of group 1 of the modern, periodic table contain two electrons in their, outermost shell., Assertion Noble gases are also called inert gases., Reason Noble gases have a complete octet., Assertion Atomic size increases on going down the, group in the modern periodic table., Reason New shells are being added as we go down, the group in the modern periodic table., Assertion Electronegativity of flourine is greater than, that of oxygen., Reason The electronegativity of the elements increases, along a period since the metallic character increases., , Case Based MCQs, 21. Read the following and answer the questions from (i), , !, , to (v) given below, The modern periodic table is based on the increasing, order of atomic radius. The atomic size increases, down the group. This is because new shell are being, added as we go down the group. The atomic radius, decreases in moving from left to right along a period., , (i) Who discovered modern periodic table?, (a) Henry Moseley, (b) Johann Wolfgang Dobereiner, (c) John Newlands, (d) Dmitri Ivanovich Mendeleev, , (ii) Along the period 3, which atom can loose electron, easily?, (a) Li, , (b) Na, , (c) Be, , (d) Mg, , (iii) Which metal has the highest metallic character in, group 1?, (a) Li, , (b) Na, , (c) K, , (d) Rb, , (iv) Which of the following properties increases down, the group?, (a) Electronegativity, (b) Electropositive nature of element, (c) Non-metallic character, (d) Acidic oxide character, , (v) Which atom is most electronegative along the, period 4?, (a) K, , (b) Rb, , (c) Sr, , (d) Ca, , 22. Read the following and answer the questions from, (i) to (v) given below, The noble gases make up a class of chemical, elements with similar properties under standard, conditions, they are all odourless, colourless,, monoatomic gases with very low chemical reactivity., These gases are also called as inert gases or, aerogens. The properties of the noble gases can be, well explained by modern theories of atomic, structure. Their outer shell of valence electrons is, considered to be full, giving them little tendency to, participate in chemical reactions., The linkage formed from one electrostatic attraction, between oppositely charged ions in a chemical, compound is called ionic or electrocovalent bond., Such a bond forms when the valence electrons of, one atom are transferred permanently to another, atom.
Page 3 :
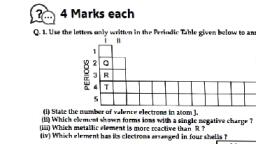
24, , CBSE Term II Science X, , The table given below refers to the elements of the, periodic table with atomic number from 3 to 18., These elements are shown by letters., (Not by the usual symbols of the elements)., 3, , 4, , 5, , 6, , 7, , 8, , 9, , 10, , A, , B, , C, , D, , E, , F, , G, , H, , 11, , 12, , 13, , 14, , 15, , 16, , 17, , 18, , I, , J, , K, , L, , M, , N, , O, , P, , (i) Which of the following are noble gases ?, (a) H and P, (c) D and L, , (b) C and M, (d) E and P, , (iii) Which of the following elements have valency 4 ?, (a) F and N, (c) D and L, , (b) C and K, (d) H and P, , (iv) Which of the following can form ionic bond?, (a) B and H, (c) E and P, , (b) J and P, (d) A and G, , (d) C and K, , PART 2, Subjective Questions, !, , (NCERT), , 7. In Mendeleev’s periodic table, the elements were, arranged in the increasing order of their atomic, masses. However, cobalt with atomic mass of, 58.93 amu was placed before nickel having an, atomic mass of 58.71 amu. Give reason for the same., (NCERT Exemplar), , 8. Write the formulae of chlorides of Eka-silicon, , 10. How it can be proved that the basic structure of the, ‘modern periodic table’ is based on the electronic, configuration of atoms of different elements?, (CBSE 2019), , 11. ‘Hydrogen occupies a unique position in modern, periodic table’. Justify the statement.(NCERT Exemplar), , (v) Which of the following elements have 2 valence, electrons?, (a) D and L (b) B and J (c) A and I, , creating his periodic table?, , and Eka-aluminium, the elements predicted by, Mendeleev., 9. Write two main characteristics of Mendeleev’s, periodic table and write name of elements of second, period., , (b) G and O, (d) A and I, , (ii) Which are halogens ?, (a) H and L, (c) G and O, , 6. What were the criteria used by Mendeleev in, , element A (atomic number 19) combines with the, element B (atomic number 17). Draw its electronic, dot structure. What is the nature of the bond, formed?, (NCERT Exemplar), , 1. What were the limitations of Dobereiner’s, (NCERT), , 2. Can the following groups of elements be classified as, Dobereiner’s triad?, (i) Na, Si, Cl, (ii) Be, Mg, Ca, Atomic mass of Be-9; Na-23; Mg-24; Si-28; Cl-35; Ca-40, Justify your answer in each case., (CBSE 2019), , 3. Did Dobereiner’s triads also exist in the columns of, Newlands’ octaves? Compare and find out., , Mendeleev’s periodic table and the modern, periodic table., (ii) With the help of an example, explain, Dobereiner’s Triads., (iii) State modern periodic law., (CBSE 2020), , 13. Write the formula of the product formed when the, , Short Answer Type Questions, classification?, , 12. (i) List any two distinguishing features between, , (NCERT), , 4. Elements have been arranged in the following, sequence on the basis of their increasing atomic, masses. F, Na, Mg, Al, Si, P, S, Cl, Ar, K., (i) Pick two sets of elements which have similar, properties., (ii) The given sequence represents which law of, classification of elements?, 5. What were the limitations of Newlands’ law of, (NCERT), octaves?, , 14. Compare the radii of two species X and Y. Give, reasons for your answer., (a) X has 12 protons and 12 electrons, (b) Y has 12, protons and 10 electrons, (NCERT Exemplar), , 15. Arrange the following elements in increasing order of, their atomic radii., (i) Li, Be, F and N, , (ii) Cl, At, Br and I, (NCERT Exemplar), , 16. An element X of group 15 exists as diatomic, molecule and combines with hydrogen at 773 K in, presence of the catalyst to form a compound,, ammonia which has a characteristic pungent smell., (i) Identify the element X. How many valence, electrons does it have?, (ii) Draw the electron dot structure of the diatomic, molecule of X. What type of bond is formed in it?, (iii) Draw the electron dot structure for ammonia and, what type of bond is formed in it?
Page 4 :
25, , CBSE Term II Science X, , Answer the following question in relation to the, above group of elements., (i) Which element has the most metallic character?, (ii) Which element would be expected to have the, highest electronegativity?, (iii) Will the elements in the group to the right of this, boron group be more metallic or less metallic in, character? Justify your answer., , 17. A salt when dissolved in water dissociates, into cations and anions as follows:, + H2 O, , AB, , !, – H2 O, , A+ + B−, , If both the ions consist same number of electrons, and the molecular weight of salt is 74.5, then identify, the position of A and B in the periodic table., , 18. Three elements A, B and C have 3, 4 and 2 electrons, respectively in their outermost shell. Give the group, number to which they belong in the modern periodic, table. Also, give their valencies., (NCERT Exemplar), , 19. Based on the group valency of elements, write the, molecular formula of the following compounds, giving justification for each., (i) Oxide of first group elements, (ii) Halide of the elements of group thirteen, (iii) Compound formed when an element A of group, 2 combines with an element, B of group seventeen., (CBSE 2019), , 20. From the elements Li, K, Mg, C, Al, S identify the, (i) elements belonging to the same group., (CBSE 2020), (ii) element which has the tendency to lose two electrons., (iii) element which prefers sharing of electrons to, complete its octet., (iv) most metallic element., (v) element that forms acidic oxide., (vi) element that belongs to group 13., , 21. The following table shows the position of five, 1, , 2, , A, , 3, , 2, , 3 to 12, , 13, , 14, , 15, , 16, , 17 18, B, , D, , C, , E, , Answer the following giving reasons:, (i) Which element is a metal with valency two?, (ii) Which element is least reactive?, (iii) Out of D and E which element has a smaller atomic, radius?, (CBSE Sample Paper), , 22. (i) What term can be used for the elements separating, metal from non-metals and why?, (ii) Give the names of the metalloids in the periodic, table along with their atomic number., (iii) In which groups of the periodic table are they, located?, , 23. A group of elements in the periodic table are given, below (boron is the first member of the group and, thallium is the last)., Boron, aluminium, gallium, indium, thallium, , Long Answer Type Questions, , 24. Use Mendeleev’s periodic table to predict the, formulae for the oxides of the following elements., K, C, Al, Si and Ba., (NCERT), , 25. Compare and constrast the arrangement of element in, Mendeleev’s periodic table and the modern periodic, table., (NCERT), , 26. An element is placed in 2nd group and 3rd period of, the periodic table, burns in presence of oxygen to, (NCERT Exemplar), form a basic oxide., (i) Identify the element., (ii) Write the electronic configuration., (iii) Write a balanced equation when it burns in the, presence of air., (iv) Write a balanced equation when this oxide is, dissolved in water., (v) Draw the electron dot structure for the formation of, this oxide., , 27. Which elements has, , elements A, B, C, D and E in the modern periodic, table., Group →, Period ↓, , !, , (i) two shells, both of which are completely filled with, electrons?, (ii) the electronic configuration 2, 8, 2?, (iii) a total of three shells, with four electrons in its, valence shell?, (iv) a total of two shells, with three electrons in its, valence shell?, (v) twice as many electrons in its second shell as in its, first shell?, (NCERT), , 28. An element X (atomic number =17 ) reacts with an, element Y (atomic number = 20) to form a divalent, halide., (i) Where in the periodic table are elements X and Y, placed?, (ii) Classify X and Y as metal(s), non-metal(s) or, metalloid(s)., (iii) What will be the nature of oxide of element Y?, Identify the nature of bonding in the compound, formed., (iv) Draw the electron dot structure of the divalent, halide., (NCERT Exemplar)
Page 5 :
26, , CBSE Term II Science X, , 29. (i) Using the part of the periodic table given below, answer the questions that follows:, Groups → 1, , 2, , 13, , 14, , 15, , 16, , 17, , 18, , (iii), , Periods ↓, 1, , H, , 2, , Li, , B, , C, , N, , O, , F, , He, Ne, , 3, , Na Mg Al, , Si, , P, , S, , Cl, , Ar, , 4, , K, , Be, Ca, , (a) Na has physical and chemical properties similar to, which element(s)., (b) Write the electronic configuration of N and P., Which one of these will be more electronegative, and why?, (c) State a chemical property common to fluorine and, chlorine., , (ii) The neutral atom of an element E consists 12, electrons in its atoms., (a) In which period and group is E placed?, (b) Name the element E., (c) How many electrons it needs to lose or gain to, achieve noble gas configuration ?, (d) What will be the nature of oxide (acidic/basic) of E?, Justify your answer., (e) Write the formulae of chloride of E., !, , (ii), , Case Based Questions, , 30. Read the following and answer the questions from (i), to (v) given below, Modern periodic law states that the physical and, chemical properties of the elements are periodic, function of their atomic radius. When these elements, were arranged in the increasing order of their atomic, number, the obtained table is called modern periodic, table., Numerous forms of the periodic table have been, devised form time to time. A new version, which is, most convinient and widely used is the modern, periodic table. This table consist of 18 vertical, columns called groups and 7 horizontal rows,, known as periods. The first period consists of two, elements., The subsequent period consist of 8, 8, 18, 18 and 32, elements respectively. The seventh period is, incomplete and like the sixth period would have, maximum of 32 electrons., (i) An element belongs to group 17. It is present in, third period and its atomic number is 17. What is, , (iv), (v), , the atomic number of the element belonging to, same group and present in fifth period ?, Name the elements present in the first period of the, modern periodic table., Atoms of different elements with same number of, shells are placed in the same period. Explain., What is the electronic configuration of the element, present in third period and belongs to group 15 ?, How many elements are present in mdoern periodic, table ?, , 31. Read the following and answer the questions from (i), to (v) given below, Valency is the combining capacity of an atom of an, element to acquire noble gas configuration. If, depends upon the number of valence electrons, present in outermost shell of its atom. For the, elements of group 1, 2, 13 and 14 valency = numbers, of valency electrons(s), whereas for the elements of, group 15 onwards valency = 8 − valence electrons., The concept of valency is simple and rationalise the, atomic composition of a large number of compounds., Yet, in many chemistry courses, it is sidelined in, favour of electronic theories of bonding, which are, more difficult. When the theory of valency was, devised, chemists thought that all compounds were, molecular. We now known that many are, non-molecular, i.e. they comprise a large number of, atoms bound together in a continuous framework., The theory can however be adapted to include, non-molecular compounds., The atomic number and valence of element A, B, C,, D and E are given in below table., Elements Atomic number Number of valence electron, A, , 3, , 1, , B, , 9, , 7, , C, , 17, , 7, , D, , 20, , 2, , E, , 36, , 0, , (i) What is the valency of element B ?, (ii) If element C reacts with an element D, which type, of compound they will form., (iii) What is the name of element E ?, (iv) Element A belongs to which period in the modern, periodic table ?, (v) Among A, C, D and E, which element belongs to, group 17 ?
 Learn better on this topic
Learn better on this topic