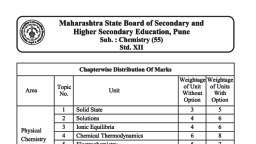
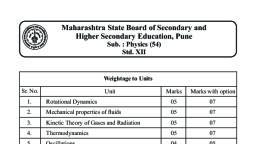
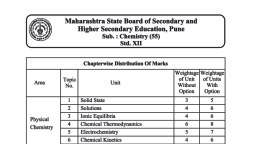
Subjective Test of 11 Th Science, Chemistry Unit Test - Study Material
Page 1 :
Samarth Junior College, Belhe, Tal-Junnar, Dist-Pune 412 410, Date:, Time-1:30 Hrs, , first Unit Test 2021-22, Chemistry, , 11thScience, Marks:-25, , General Instructions: The question paper is divided into four sections., 1)Section A:Q.No.1 contains Ten multiple choice type of questions carrying one, mark each.Q.No.2 contains Eight very short answer type of questions carrying, one mark each., 2)Section B:Q.No.3 to Q.No.14 contains Twelve short answer type of questions, carrying Two marks each., 3)Section C:Q.No.15 to Q.No.26 contains Twelve short answer type of questions, carrying Three marks each., 4)Section D:Q.No.27 to Q.No.31 contains five long answer type of questions carrying four marks each., 5)Use of log-table if allowed. Use of calculator is not allowed., 6)Figures to the right indicate full marks, 7)For each MCQ correct answer must be written along with it's alphabet., e.g.(a)....................(b)22222..(c)222222.(d)222222, 8)Physical constants:, Molar gas constants’=8.314 J K-1 mol-1Avogadro's constant=6.022x 1023, ............................................................................................................................................................, , Section A, Q.1 Select and write the correct answer:, , 4M, , I The number of significance figure in 1.15X104 gm is ……………….., a. 2, b.3, c.4, d.6, II Which of the following techniques is used for separation of glycerol from soap in soap, industry?, a.Distillation under reduced pressure, , b.Fractional distillation, , c.Filtration, , d.Crystallization, , III. A Simple of pure water, whatever the source always contains ................... by mass, of oxygen and 11.1% by mass of hydrogen., a.88.9, b. 18, c.80, d.16, IV SI unit of the the quantity electric current is....., a.Volt, , b.Ampere, , c.Candela, , d.Newton
Page 2 :
Q.2 Answer the following:, , 3M, , I Define stationary phase., II Define analytical chemistry., III State the law of multiple proportion., ………………………………………..Section B, Attempt any Four, , 8M, , Q.3 State and explain Avogadro’s law., Q.4 How many particles are present in 1 mole of a substance., Q.5 Distinguish between accuracy and precision., Q.6. How many significant figures are in each following quantities., I. 0.00025, II 45.26, Q.7 Define the terms residue and filtrate., , Q.8 Draw &Label the diagram of filtration under suction., Section C, Attempt any Two, 6M, Q.9 Calculate the mass of carbon dioxide and water formed on complete combustion of, 24 gram of methane gas.(Given atomic mass C= 12u ,H= 1u,O=16u), Q.10 Difference between simple distillation & fractional distillation?., Q.11Calculate molecular mass of the following in u., , a)NH3, , b) CH3COOH, , c) C2H5OH, Section D, , Attempt any One, Q.12 Define compound and element., Calculate the following degree Celsius temperature to degree Fahrenheit., , 4M, , a)40OC, b)30OC, Q.13 Explain the molality., A substance on analysis of the following percent composition Na= 43.4 %, C= 11.3%, and O= 45.3%.Calculate the empirical formula (Na= 23u, C= 12 u,O= 16u.)
 Learn better on this topic
Learn better on this topic