Notes of Plus One, Chemistry s block.pdf - Study Material
Page 1 :
10. S-BLOCK ELEMENTS, HAIZEL G. ROY, H.S.S.T. (HG) CHEMISTRY, GOVT. H.S.S. KALAMASSERY, ERNAKULAM
Page 2 :
In, , s-block elements the last electron enters the outermost, , s-orbital., It includes elements of group 1 and 2 and He in group 18., It consists of six elements Li, Na, K, Rb, Cs and Fr., These, , are called alkali metals because they react with, , water to form soluble hydroxides which are strong bases or, alkalies.
Page 3 :
The group 2 includes Be, Mg, Ca, Sr, Ba and Ra., These, , elements with the exception of Be are commonly, , known as alkaline earth metals., They, , are so called because their oxides are found in the, , earth’s crust., The, , general outer electronic configuration of s-block, , elements is ns1 or ns2 .
Page 4 :
DIAGONAL RELATIONSHIP, The, , similarity in properties shown by diagonally placed, , elements of second and third periods in modern, periodic table is called diagonal relationship., Li shows similarities to Mg., Be shows similarities Al.
Page 5 :
CAUSE OF DIAGONAL RELATIONSHIP, As, , we move from left to right across a period, the electronegativity, , increases., But it decreases when we move from top to bottom., As, , a result of these two opposite changes, as we move diagonally,, , these two effects tend to cancel each other and there is no marked, change in electronegativity., Because, , of the similar values of electronegativities, the diagonal, , elements have similar chemical properties.
Page 6 :
ANOMALOUS BEHAVIOUR OF THE FIRST MEMBER OF A GROUP, The, , first member in each group shows certain properties which are, , different from that of the other elements in their respective groups., This anomalous behaviour of the first member in each group is due to, The very small size of the atoms, High Ionisation energies, High electronegativities, Absence of vacant d orbitals.
Page 7 :
GROUP I ELEMENTS, ALKALI METALS
Page 8 :
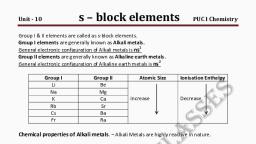
ATOMIC AND PHYSICAL PROPERTIES, ELECTRONIC CONFIGURATION, , The general electronic configuration of alkali metals, may be represented as [Noble gas] ns1 .
Page 9 :
ATOMIC AND IONIC RADII, The alkali metals have the largest sizes in a particular period, , of the periodic table., With, , the increase in atomic number, the atom becomes, , larger., The monovalent ions (M+) are smaller than the parent atom., The, , atomic and ionic radii of alkali metals increase on, , moving down the group.
Page 10 :
IONISATION ENERGY, The alkali metals have low ionisation energies., Their atomic sizes are quite large and the valence electrons are, , quite loosely held by the nucleus., On moving down the group, the ionisation energy decreases., It is due to the increase in size of the atoms., Increase, , in the magnitude of screening effect caused by the, , increase in the number of intervening electrons.
Page 11 :
HYDRATION ENTHALPY, The, , hydration enthalpies of alkali metal ions decrease, , with increase in ionic sizes., The smaller the ion, the more is the extent of hydration., Due, , to this, in aqueous solution, the hydration enthalpy, , decreases in the order Li+ > Na+ > K+ > Rb+ > Cs+
Page 12 :
PHYSICAL PROPERTIES
Page 13 :
1. METALLIC CHARACTER, All, , the alkali metals are silvery white, soft and light, , metals., The metallic character increases down the group., 2. DENSITY, Because of the large size, alkali metals have low density., The Density generally increases down the group.
Page 14 :
MELTING AND BOILING POINTS, The melting and boiling points of alkali metals are low., It, , indicates weak metallic bonding due to the presence of, , only a single valence electron in them., The, , melting and boiling points decreases down the, , group.
Page 15 :
When alkali metals or their salts are heated in the flame of a Bunsen, , burner, they impart characteristic colours to the flame.
Page 16 :
Alkali metals have low Ionisation energy., Therefore,, , the electrons present in their atoms get readily, , excited to higher energy states by absorbing energy from the, visible region of the light., When, , these electrons jump back to their ground state, they, , emit energy in the form of radiations., These, , radiations fall in the visible region, imparting, , characteristic colouration to the flame.
Page 17 :
CHEMICAL PROPERTIES
Page 18 :
REACTIVITY TOWARDS AIR, Alkali metals tarnish in dry air., It is due to the formation of their oxides., These oxides in turn react with moisture to form, , hydroxides., They burn vigorously in oxygen forming oxides.
Page 19 :
When heated in excess of air, Li forms normal oxide., Sodium forms peroxides., K, Rb and Cs forms superoxide.
Page 20 :
REACTIVITY TOWARDS WATER, The alkalie metals react with water to form hydroxide, and dihydrogen.
Page 21 :
REACTIVITY TOWARDS DIHYDROGEN, On, , heating, alkali metals react with hydrogen to form hydrides of the, , formula, MH., All the alkali metal hydrides are ionic solids with high melting points., , The hydrides are strong reducing agents., It reduce water with the liberation of hydrogen.
Page 22 :
REACTIVITY TOWARDS HALOGENS, Alkali metals react with halogens to form halides of the, general formula, MX.
Page 23 :
REDUCING NATURE, The alkali metals are strong reducing agents., Lithium, being the most and sodium, the least powerful., The, , reducing character increases down the group from, , Na to Cs because of the decrease in ionisation energy.
Page 24 :
SOLUBILITY IN LIQUID AMMONIA, The, , alkali metals dissolve in liquid ammonia giving deep blue, , solutions which are conducting in nature., The, , blue colour of the solution is due to the ammoniated electrons, , (solvated electrons)., Ammoniated electrons absorbs energy in the visible region of light., This imparts blue colour to the solution., The solutions are paramagnetic.
Page 25 :
SALTS OF OXO ACIDS, Since, , alkali metals are strongly electropositive, they, , form salts with oxo acids., Oxo, , acids are those in which the acidic proton is on a, , hydroxyl group with an oxo group attached to the same, atom.
Page 27 :
ANOMALOUS PROPERTIES OF LITHIUM, Lithium is much harder., The, , melting point and boiling point are higher than the, , other alkali metals., Lithium, , is the least reactive but the strongest reducing, , agent among all the alkali metals.
Page 28 :
Lithium reacts with nitrogen to form a nitride, Li, , 3, , N., , LiCl is deliquescent and crystallizes as a hydrate, LiCl. 2H, , 2, , O., , LiOH is a weak base., Lithium does not form acetylide on reaction with acetylene.
Page 29 :
DIAGONAL SIMILARITIES BETWEEN Li and Mg, Both, , Li and Mg are harder than other elements in the, , respective groups., Both, , Li and Mg form a nitride by direct combination with, , nitrogen, Both, , Li and Mg react with oxygen to form normal oxides, , Li2O and MgO respectively.
Page 30 :
The, , hydroxides of Li and Mg are weak bases and they, , decompose on heating., Carbonates, , of Li and Mg decompose easily on heating to, , form the oxide and CO2 ., Both LiCl and MgCl, Both, , 2, , are soluble in ethanol., , LiCl and MgCl2 are deliquescent and crystallizes as, , hydrates LiCl.2H2O and MgCl2 .8H2O.
Page 31 :
SOME IMPORTANT, COMPOUNDS OF SODIUM
Page 32 :
Sodium, , forms many industrially important compounds, , like, sodium carbonate (Na, , 2, , CO3), , sodium chloride (NaCl), sodium hydroxide (NaOH), sodium hydrogen carbonate (NaHCO, , 3, , )
Page 33 :
SODIUM CARBONATE (Na2CO3), Sodium carbonate is also known as washing soda or soda, ash.
Page 34 :
PREPARATION, Sodium carbonate is generally prepared by Solvay process., Prepared, , by passing CO2 gas under pressure into a strong sodium, , chloride solution saturated with ammonia., Sodium Hydrogen Carbonate is precipitated.
Page 35 :
Sodium hydrogen carbonate on heating gives sodium carbonate., , In this process, NH, , 3, , is recovered when the solution containing NH4Cl, , is treated with Ca(OH)2 ., Calcium chloride is obtained as a by-product.
Page 36 :
K2CO3CANNOT BE PREPARED BY SOLVAY PROCESS, Potassium, , carbonate cannot be prepared by Solvay, , process., This is because the solubility of KHCO, Hence, , tower., , 3, , is fairly large., , it does not precipitate easily in the carbonation
Page 37 :
PROPERTIES, Sodium carbonate is a white crystalline solid., It exists as a decahydrate, Na, , 2, , CO3 .10H2O., , This is also called washing soda., It is readily soluble in water., On, , heating, the Decahydrate loses its water of crystallization, , to form monohydrate.
Page 38 :
Above, , 373K,, , the, , monohydrate, , becomes, , completely, , anhydrous and changes to a white powder called soda ash., , The solution of Na, , 2, , CO3 is alkaline due to hydrolysis.
Page 39 :
USES, Sodium Carbonate is used, in water softening, laundering and cleaning., in the manufacture of glass, soap, borax and caustic soda., in paper, paints and textile industries., It, , is an important laboratory reagent both in qualitative and, , quantitative analysis.
Page 40 :
SODIUM CHLORIDE (NaCl), The most abundant source of sodium chloride is sea water., Sea water contains 2.7 to 2.9 % by mass of the salt.
Page 41 :
PREPARATION, In India, common salt is generally obtained by the evaporation of sea, , water., Crude NaCl is generally obtained by crystallization of brine solution., It, , contains sodium sulphate, calcium sulphate, calcium chloride and, , magnesium chloride as impurities., To, , obtain pure sodium chloride, the crude salt is dissolved in, , minimum amount of water.
Page 42 :
The insoluble impurities are filtered., The, , solution is then saturated with hydrogen chloride, , gas., Crystals of pure sodium chloride separate out., Calcium, , and magnesium chloride being more soluble, , than sodium chloride, remain in solution.
Page 43 :
PROPERTIES, Sodium chloride melts at 1082K., It has a solubility of 36g in 100g of water at 273K., The, , solubility does not increase appreciably with increase, , in temperature.
Page 44 :
USES, It, , is used as common salt or table salt for domestic, , purpose., It is used for the preparation of Na, , 2, , O2 , NaOH and Na2CO3 .
Page 45 :
SODIUM HYDROXIDE (NaOH), Sodium hydroxide is also known as caustic soda.
Page 46 :
PREPARATION, Sodium, , hydroxide is prepared commercially by the electrolysis of, , sodium chloride in Castner-Kellner cell., Sodium hydroxide is obtained by the electrolysis of brine solution., Cathode is made up of mercury and anode is made up of carbon., Sodium metal is discharged at the cathode., It combines with mercury to form sodium amalgam., Chlorine gas is evolved at the anode.
Page 47 :
The amalgam is treated with water to give sodium hydroxide, and hydrogen gas.
Page 49 :
PROPERTIES, Sodium hydroxide is a white, translucent solid., It melts at 591K., It is readily soluble in water to give a strong alkaline solution., Crystals of sodium hydroxide are deliquescent., The, , sodium hydroxide solution at the surface reacts with the, , CO2 in the atmosphere to form Na2CO3 .
Page 50 :
USES, Sodium hydroxide is used: in the manufacture of paper, soaps, artificial silk etc., for the purification of bauxite., as an important laboratory reagent., in petroleum refining., for mercerizing cotton fibres., for refining vegetable oils., for preparing various sodium salts like sodium chlorate, hypochlorite, , etc.
Page 51 :
SODIUM HYDROGEN CARBONATE, Sodium Hydrogen Carbonate is known as baking soda., It, , is called so because it decomposes on heating to generate bubbles, , of CO2 .
Page 52 :
PREPARATION, It, , is prepared by saturating a solution of sodium carbonate, , with carbon dioxide., The, , white crystalline powder of sodium hydrogen carbonate,, , being less soluble, gets separated out.
Page 53 :
USES, NaHCO, , 3, , is used, , as a laboratory reagent., in baking powder., in fire extinguishers., as, , a mild antiseptic for skin, , infections.
Page 54 :
BIOLOGICAL IMPORTANCE OF SODIUM, Sodium ions are found outside the cells., It, , is located in blood plasma and in the interstitial fluid which, , surrounds the cells., These ions participate in the transmission of nerve signals., Regulates the flow of water across cell membranes, Helps in the transport of sugars and amino acids into cells.
Page 55 :
BIOLOGICAL IMPORTANCE OF POTASSIUM, Potassium, , ions are the most abundant cations within cell, , fluids., They activate many enzymes., Participate in the oxidation of glucose to produce ATP., With, , sodium it is responsible for the transmission of nerve, , signals.
Page 56 :
GROUP II ELEMENTS, ALKALINE METALS
Page 57 :
Group, , 2 of the periodic table consists of the elements Be, Mg,, , Ca, Sr, Ba and Ra., These, , elements except beryllium are known as alkaline earth, , metals., Radium, the last member of the group is radioactive in nature., Beryllium shows diagonal relationship to Aluminium.
Page 58 :
ATOMIC AND, PHYSICAL PROPERTIES
Page 59 :
ELECTRONIC CONFIGURATION, The, , general electronic configuration may be, , represented as [Noble gas] ns2, where n = 2 to 7.
Page 60 :
ATOMIC RADII, The, , atomic and ionic radii of the alkaline earth metals are, , smaller than those of the corresponding alkali metals in the, same periods., This is due to the increased nuclear charge in these elements.
Page 61 :
IONISATION ENERGIES, The, , alkaline earth metals have low ionisation energies due to, , fairly large size of the atoms., The, , first ionisation energies of the alkaline earth metals are, , higher than those of the corresponding group I metals., This, , is due to their small size as compared to the, , corresponding alkali metals.
Page 62 :
HYDRATION ENTHALPIES, The hydration enthalpies of alkaline earth metal, , ions decrease, , with increase in ionic size down the group., Be2+ > Mg2+ > Ca2+ > Sr2+ > Ba2+, The, , hydration enthalpies of alkaline earth metal ions are, , larger than those of alkali metal ions., Compounds, , of alkaline earth metals are more extensively, , hydrated than those of alkali metals.
Page 63 :
PHYSICAL, PROPERTIES
Page 64 :
METALLIC CHARACTER, The alkaline earth meals are silvery white, lustrous and, , relatively soft, but harder than the alkali metals., Be and Mg appears to be somewhat greyish.
Page 65 :
DENSITY, The, , alkaline earth metals are denser and harder than the, , alkali metals of the corresponding periods., It, , is because of the smaller size and stronger metallic bonds, , resulting in more closely packed crystal lattices.
Page 66 :
MELTING AND BOILING POINTS, Alkaline earth metals have higher melting and boiling points., This, , is because of their smaller size and more closely packed, , structures.
Page 67 :
ELECTROPOSITIVE CHARACTER, Alkaline earth metals are electropositive in nature., Due, , to, , higher, , ionisation, , energies,, , these, , are, , less, , electropositive than alkali metals., Within the group, the electropositive character increases from, , Be to Ba.
Page 68 :
FLAME COLOURATION, Ca,, , Sr and Ba impart characteristic brick red, crimson red and, , apple green colours to the flame respectively., Be, , and Mg does not impart any colour to the flame due to, , small size and high ionisation energies., The, , excitation of electrons is not possible by the energy, , available from the flame.
Page 69 :
CHEMICAL, PROPERTIES
Page 70 :
REACTIVITY TOWARDS AIR, Be, , and Mg are kinetically inert to oxygen and water because of the, , formation of an oxide film on their surface., Powdered Be burns brilliantly on ignition in air to give BeO and Be, Mg, , N2 ., , 3, , is more electropositive and burns with dazzling brilliance in air to give, , MgO and Mg3N2 ., Ca, Sr and Ba are readily attacked by air to form the oxide and nitride., They, , also react with water with increasing vigour even in cold to form, , hydroxides.
Page 71 :
REACTIVITY TOWARDS HYDROGEN, All, , the elements except Be combine with hydrogen upon, , heating to form their hydrides, MH2., , BeH, , 2, , can be prepared by the reaction of BeCl2 with LiAlH4.
Page 72 :
REACTIVITY TOWARDS ACIDS, The alkaline earth metals readily react with acids liberating, dihydrogen.
Page 73 :
REACTIVITY TOWARDS HALOGENS, All, , the alkaline earth metals combine with halogen at, , elevated temperatures forming their halides., BeCl, , 2, , is formed by passing chlorine over a heated mixture of, , Beryllium oxide and carbon.
Page 74 :
STRUCTURE OF BeCl2, In the solid state, BeCl, , 2, , exists as a polymer., , Each Be atom is surrounded by four chlorine atoms., Two, , of the chlorine atoms are bonded by covalent bonds and, , the remaining two by coordinate bonds.
Page 75 :
REDUCING NATURE, Alkaline earth metals are strong reducing agents., They have large negative values of their reduction potentials., Their, , reducing power is less than those of their corresponding, , alkali metals.
Page 76 :
SOLUTIONS IN LIQUID AMMONIA, Alkaline earth metals dissolve in liquid ammonia to give deep, blue black coloured solutions forming ammoniated ions.
Page 77 :
FORMATION OF OXIDES, Alkaline, , earth metals react with air or oxygen slowly upon, , heating to form oxides., Be, Mg and Ca forms monoxides., Sr and Ba forms peroxides.
Page 78 :
FORMATION OF HYDROXIDES, Elements of group 2 have lesser tendency to react with water., On, , heating, they react with water to form hydroxide and, , liberate hydrogen., The, , hydroxides are basic in nature, except Be(OH)2 which is, , amphoteric., The basic strength increases down the group., It is due to the decrease in the ionisation energies.
Page 79 :
ANOMALOUS BEHAVIOUR OF BERYLLIUM, The properties of Be differs from those of the other elements, , of the group., It is because of, Its small size, Relatively high ionisation energy, Relatively high electronegativity, Absence of vacant d orbitals in the valence shell.
Page 80 :
DIFFERENCES, Be compounds are more covalent in nature., Be does not react with water even at high temperatures., BeO is amphoteric., Be does not react with H, , 2, , Be, , 2, , to give hydride., , C reacts with water to give methane.
Page 81 :
DIAGONAL RELATIONSHIP BETWEEN Be and Al, Both Be and Al have same electronegativity., Both Be and Al form covalent compounds., Both Be and Al are resistant to the action of acids., The hydroxides of Be and Al are amphoteric., Chlorides, , of both Be and Al have bridged structures in vapour, , phase., Carbides, , methane., , of both Be and Al undergo hydrolysis to give
Page 82 :
SOME IMPORTANT COMPOUNDS, OF CALCIUM
Page 83 :
COMPOUNDS OF CALCIUM, Important compounds of calcium are, Calcium oxide, Calcium hydroxide, Calcium sulphate, Calcium carbonate, Cement
Page 84 :
CALCIUM OXIDE OR QUICK LIME (CaO)
Page 85 :
PREPARATION, It, , is prepared on a commercial scale by heating limestone in a, , rotary kiln at 1070-1270K., , The, , CO2 is removed as soon as it is produced to enable the, , reaction to proceed to completion.
Page 86 :
PROPERTIES, Calcium oxide is a white amorphous solid., It has a melting point of 2870K., On, , exposure to atmosphere, it absorbs moisture and carbon, , dioxide., The, , addition of limited amount of water breaks the lump of, , lime., This process is called slaking of lime.
Page 87 :
USES, Calcium Oxide is used, as a building material, in softening of hard water., in the manufacture of calcium carbide., as a drying agent., in the purification of sugar.
Page 88 :
CALCIUM HYDROXIDE OR SLAKED LIME Ca(OH)2
Page 89 :
PREPARATION, Calcium hydroxide is prepared by adding water to quick lime,, CaO.
Page 90 :
PROPERTIES, Calcium hydroxide is a white amorphous powder., It is sparingly soluble in water., A suspension of slaked lime in water is called milk of lime., The filtered solution is called lime water.
Page 91 :
When, , CO2 is passed through lime water, it turns milky due to, , the formation of CaCO3 ., On, , passing excess of CO2 , the precipitate dissolves to form, , calcium hydrogen Carbonate., Milk, , of lime reacts with chlorine to form hypochlorite, a, , constituent of bleaching powder.
Page 92 :
USES, Calcium Hydroxide is used, in the production of mortar, a building material., as lime water in laboratories., for the manufacture of bleaching powder., Milk of lime is used for white washing.
Page 93 :
CALCIUM CARBONATE (CaCO3), It occurs in nature in several forms like limestone, chalk,, marble etc.
Page 94 :
PREPARATION, Prepared by passing carbon dioxide through slaked lime or by, the addition of Na2CO3 to CaCl2 .
Page 95 :
PROPERTIES, Calcium carbonate is a white fluffy powder., It is almost insoluble in water., When heated to 1200K, it decomposes to evolve carbon, , dioxide., It reacts with dilute acids to liberate carbon dioxide.
Page 96 :
USES, Calcium Carbonate is used, as a building material in the form of marble., for the manufacture of quick lime., Calcium carbonate along with MgCO, Specially precipitated CaCO, , 3, , 3, , is used as a flux in metallurgy., , is extensively used in the manufacture of, , high quality paper., as, , an antacid, mild abrasive in tooth paste, a constituent of chewing, , gum and filler in cosmetics.
Page 98 :
PREPARATION, Plaster of Paris is obtained by heating gypsum to 393 K., , Above, , 393 K, no water of crystallization is left and anhydrous, , calcium sulphate, CaSO4 is formed., This is known as dead burnt plaster.
Page 99 :
When, , plaster of Paris is mixed with water, it forms a paste, , which sets to a hard mass in 5 to 15 min., This is called setting of Plaster of Paris., During setting, it takes up water and forms CasO, , 4, , .2H2O.
Page 100 :
USES, Calcium Sulphate is used, for setting broken and fractured bones., for making statues, models, decorative materials etc., for producing moulds for Industries like pottery and ceramics., in dentistry and in ornamental work.
Page 101 :
CEMENT, Cement, , is an important building, , material., It, , was first introduced in England in, , 1824 by Joseph Aspidin., It is also called Portland cement., It, , resembles the natural lime stone, , quarried in the Isle of Portland,, England.
Page 102 :
WHAT IS CEMENT ?, Cement, , is essentially a finely ground mixture of calcium and, , aluminium silicates along with small quantities of gypsum, which sets to a hard mass when reacted with water., Cement, , is a product obtained by combining a material, rich in, , lime, CaO with other material such as clay which contains, silica, SiO2 along with the oxides of aluminium, Iron and, Magnesium.
Page 103 :
MANUFACTURE OF CEMENT, The, , raw materials for the manufacture of cement are limestone and, , clay., When clay and lime are strongly heated together, they fuse and react, , to form cement clinker., This clinker is mixed with 2-3% by weight of gypsum to form cement., Thus, , important ingredients present in Portland cement are, , dicalcium silicate (Ca2SiO4) 26%, tricalcium silicate (Ca3SiO5 ) 51%, and tricalcium aluminate (Ca3Al2O6 ) 11%.
Page 104 :
SETTING OF CEMENT, When, , cement is mixed with water it reacts to form a gelatinous mass, , which sets to a hard mass., The, , transition of cement from the gelatinous mass to a hard mass is, , called setting of cement., During setting, three dimensional cross links are formed between, ----Si―O―Si----, , and ----Si―O―Al---- chains making the material quite, , hard., The, , purpose of adding gypsum is to slow down the process of setting, , of the cement so that it gets sufficiently hardened.
Page 105 :
USES OF CEMENT, Cement is used, in concrete and reinforced concrete., in, , plastering and in construction of, , bridges, dams and buildings.
Page 106 :
BIOLOGICAL IMPORTANCE OF Mg & Ca, An adult body contains about 25g of Mg, 1200g of Ca, 5g of Iron, , and 0.06g of copper., The, , daily requirement in the human body has been estimated, , to be 200-300mg., All, , enzymes that utilize ATP in phosphate transfer require, , magnesium as the cofactor.
Page 107 :
The main pigment for the absorption of light in plants is, chlorophyll which contains Mg., About 99% of Ca is present in bones and teeth., It also plays important roles in neuromuscular function,, Interneuronal transmission, blood coagulation etc.
 Learn better on this topic
Learn better on this topic