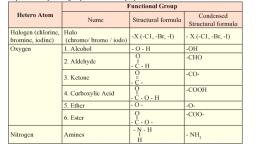
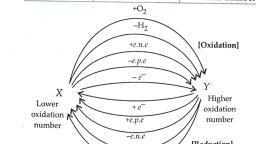
Notes of Class 11th, Chemistry Question Bank - Study Material
Page 1 :
As it rises in the air external pressure, decreases and the volume of the gas, increases till finally it bursts when, external pressure is 0.3bar. What is the, limit at which volume of the balloon can, stay inflated ? (Ans:7.567L), B. A syringe has a volume of, 10.0 cm? at pressure 1 atm. If, you plug the end so that no gas, can escape and push the plunger, down, what must be the final, volume to change the pressure, to 3.5 atm?, (Ans:2.9 cm‘), C. The volume of a given mass of a gas at, 0°C is 2 dm*. Calculate the new volume, of the gas at constant pressure when, a. The temperature is increased by 10°C., (Ans: 2.07 dm*), b.The temperature is decreased by10°C., (Ans:1.93 dm’), D. A hot air balloon has a volume of 2800, m? at 99°C. What is the volume if the, air cools to 80° C?, (Ans:2657 m*), , , , ull, , E. At 0°C, a gas occupies 22.4 liters. How, nuch hot must be the gas in celsius, and in kelvin to reach volume of 25.0, literes?, , (Ans:31.7 °C/304.9 K), , F. A 20 L container holds 0.650 mol of, He gas at 37°C at a pressure of 628.3, bar. What will be new pressure inside the, container if the volume is reduced to 12, L. The temperature is increased to 177, °C and 1.25 mol of additional He gas, was added to it?, , (Ans:4443 bar/5.85atm), , G. Nitrogen gas is filled in a container, of volume 2.32 L at 32°C and 4.7 atm, pressure. Calculate the number of moles, of the gas., , (Ans:0.436 moles), , H. At 25 °C and 760 mm of Hg pressure, a gas occupies 600 mL volume. What, will be its pressure at the height where, temperature is 10 °C and volume of the, gas 640 mL ?, , (Ans : 676.6 mm Hg), , I. A neon-dioxygen mixture contains 70.6 g, dioxygen and 167.5g neon.If pressure of, the mixture of the gases in the cylinder, is 25 bar. What is the partial pressure of, dioxygen and neon in the mixture?, , (Ans:p,= 5.25 bar, py.=19.75 bar), , J. Calculate thé pressure in atm of 1.0, mole of helium in a 2.0 dm’ container, at 20.0°C., , (Ans:12.02 atm), , K. Calculate the volume of 1 mole of a gas, at exactly 20 °C at a pressure of 101.35, kPa., , (Ans: 24.055 dm’), , L. Calculate the number of molecules of, methane in 0.50 m* of the gas at a, pressure of 2.0x10? kPa and a temperature, of exactly 300 K., , Perform different activities related to, concepts mentioned in chapter., , (Ans: 2.4x10°)
 Learn better on this topic
Learn better on this topic