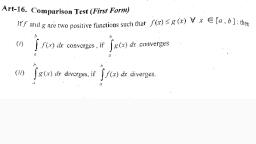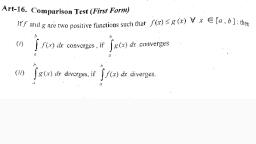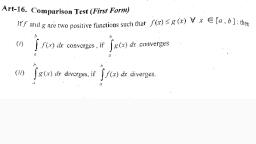Notes of BSc FY 2021-22, Chemistry & Physical Chemistry & Inorganic Chemistry Chem-P-II winter 2020.pdf - Study Material
Page 1 :
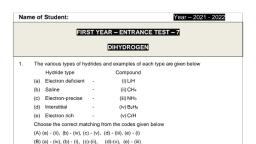
$G-5-FW2020/SG-6-FW2020, , SRTMU Nanded, FACULTY OF SCIENCE, B, Se. (First Semester) EXAN, Winter-2020, , , , Subject’ CHEMISTRY (CBCS/CRCS NEW Pattern), , Paper Title & Paper number: PHYSICAL & INORGANIC CHEMISTRY (P-ID), , Date: 07/04/2021 Time: 02.00 pm to 03.00 pm, , Time-1 Hour Maximum Marks-40, , N.B-- L_All questions are compulsory, 2 Each question carries one mark, , 1. The value of log (m/n) is, (a) log m/logn (b) log m-logn, (c) log m+logn (d) log mx log n, 2. pH ts defined as, (a) — log 10 [H+] (b) - log e [OH-], (c)— log e [H+] (d) - log 10 [OH- }, 3. The pH of the 0.00] M HCI is, , , , , , (a)0.001 —(b)3 (c) 10” (d) -3, 4. Combination of n different things taken r at a time is given by, r! nt nt : r!, @ om Om O Gain @ Bait, , 5. The derivative of e* isOR = e*, , (a) e* (b)x™ (c)nx" (d)nx™, , 6. Log jo (10000) is equal to, , (a)2 (b) 3 (c)4 (4), 7. Which of the following is incorrect, , (a) chemisorptions is caused by a chemical bond fotmation, , (b) chemisorption is specific in nature, , (c) chemisorptions is reversible, , (d) chemisorptions is unimolecular shick, 8. The adsorption in which the molecule of adsorbate are held at the surface of adsorbent by, weak vander waal's forces is called, , (a) chemical adsorption (b) physical adsorption, , (c) absorption (d) none of these., , 1[Page, , Scanned with CamScanner
Page 2 :
SG-5-FW2020/5G-6-).2020, 9, Adsorption takes place with, , (a) decrease in enthalpy of the system (b) increase in enthalpy of the system, , (c) no change in enthalpy of the system (d) none of these, 10, Which of the following ts not an equation for Freundlich isotherm, , wySeK ov" (b)Z=K pin, , (c) log — lox K + lop , (d) log <= log kK - * log P, 11, The efficiency of adsorbent increase with increase in, , (a) surface tension (b) viscosity, , (c) surface area (d) number of sons, 12. Langmuir isotherm holds at low pressure but fails at, , (a) low temperature (b) high temperature, , (c) high pressure (d) none of these, , 13. The kinetic gas equation is given by the relation, (a) PV = imnv? (b) PV = Smnv?, (c) PY = mn ve (Wd) PV = =mnv?, , 14. The value of R the gas constant is, (a) 8.314 joule k"! mole“! (b) 0.0821 litatm k! mole |, , (c) 2 cal k! mole 7 (d) All of these, , 15. The average velocity of gas molecules is given by the relation, (a) Vay= YBRT/TIM (b) Voy = V3RT/M, (c) Vv = J2RT/M— (d) Vx = RT /M, , 16. The compressibility factor 'Z’ is given by, , wl gall yZatt 72h, (a)Z= a9 (b) 2 =F ()2 = oF (d) 2= "5, 17. The Vander Waals equation explain the behavior of, (a) ideal gases (b) real gases (c) vapour —(d) non real pases., , 18. A postulate of kinetic theory 1s, , (a) atom is indivisible (b) gases combine in simple ratio by volume, , (c) there is no influence of gravity on the gas molecules (d) none of these, , 19. Critical temperature of CO) is, (a) 50°C (b)3LI"C (e)2H°C (WH) IRC, 20, The correct value of Ve is given by’ term:, , (a) 3b (b) (5 (d) None of these, , 2[eeee, , Scanned with CamScanner
Page 3 :
. : SG-5-FW2020/SG-6-FW2020, 21. Bragp's equation is given by, , (a) n= 2d sind (b) n= 20 sind, (c) 2nk= 2dsind (a) n0/2= d/2 sink, 22. The structure of KCI is, (a) body centered cubic lattice (b) face centered cubic lattice, (ce) square planer (d) octahedral, 23. NaClisa, (a) covalent erystal (b) metadic crystal, (c) molecular crystal (d) ionic crystal, 24 The number of atoms ina face centered cubic unit cell are, (a) 6 (b) 4 (c) 12 (d) 8, 25. In solids, the constituent particles may be, , (a) aloms (b) ions (c) molecules (d) either of the above three, , 26. Miller indices are denoted by, (aya, boc (b) A, kf (c)4 mn (d) None of these., 27. Face centred cubic lattice also known as, (a) simple unit cell (b) multiple unit cell, (c) cubic close packing (d) None of these, 28. The gencral electronic configuration of alkali metal is, (a) ns! (b) ns? (c) ns*np! (d) ns*np”, 29 Diagonal relationship is shown by the elements, (a) Be and Mg (b) Li and Mg (c) Li and Na (d) Be and Ba, , 30. Which is inost basic in character, (a) RbOH (b) KOH (c) LiOH (d) NaOH, , 31. The most clectropositive amongst the alkaline metal is, , (a) Be (b) Mg (c) Ca (d) Ba, 32. Which of the following metal present In chlorophyll, (a) My: (b) Be (c) Ca (d) Zn, , 33. Alkaline earth (A) compounds are less soluble in water than the alkali metals, , (IA)because alkaline carth metals have, (a) lover liltice energy (b) higher ionization energies, (c) higher covalent character (d) lower covalent character., 34. Which of the alkaline earth metal hydroxide is least alkaline, , (a) BaOI, — (b) Ca(Ol)2 (c) SOM): ~— (d) Be(ON)z, , Scanned with CamScanner
 Learn better on this topic
Learn better on this topic