Notes of 11 Sci B, Chemistry-1 IMG-20211026-WA0001.jpg - Study Material
Page 1 :
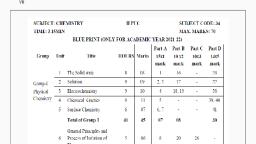
XI Chemistry (55), Deleted and Non-Evaluative portion for Year 2020-21., , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , , ‘Sr.No | Chapter No. and Name Deleted and Non-Evaluative portion, T | 1Some basic concepts of Tlintroduction 1.2 Nature of chemistry ,1-2.1 Matter, 1.2.2 Pure substances verses mixtures, 2.3 States of, chemistry ‘matter, 1.4.1 Law of conservation of mass, 1.4.2 Law of definite proportion, 1.4.3 Law of multiple proportion,, 1.5 Daltons atomic theory., 2 | Zintroduction to Analytical | 2.1 Introduction, 2.3 Mathematical operation and error analysis, 2.3.2 Scientific notation, 2.3.3 Significant, chemistry figures, 2.3.4 Rules of deciding significant figures, 2.3.5 Calculations with significant figures., 3__ | 3Some Analytical techniques | 3.1 Introduction,3.5 Chromatography, 4_[4 Structure of Atom 41 Subatomic particles, 4.5.1Wave particle duality of electromagnetic radiation (including problems 4.4 & 45)., 3__|5. Chemical Bonding 3:1 Introduction, 5.2 Kossel and Lewis approach to chemical bonding, 5.2.1 Tonic bond, 5.1.2 Tonic solids and, lattice enthalpy .5.2.2 Covalent bond, 5.4.1 Formal charge, 5.5.6.2, 5.5.6.3, 5.5.6.4.5.5.6.5 (Except H molecule), © _| 6 Redox reactions 6.1 Introduction, 6.1.1 Classical ideas of redox reactions, 63.2 Ton electron method (Half reaction method), 7 | 7-Modern Periodic Table 77.1 Introduction, 7.5.3.¢. Periodic trends in Chemical Reactivity., 8 | 8Elements of group | and2 _ | 8.1Hydrogen (Entire ),8.2.7 Biological importance of elements of group 1 and 28.3.1 Sodium Carbonate, 83.3, Calcium Carbonate, 8.3.4 Hydrogen peroxide., ‘9 | 9. Elements of group 13, 14 | 9.1 Introduction , 9.6.1 Allotropes of Carbon , 9.8 Chemistry of notable compounds of elements of group 13 ,, and 15 14 and 15, TO _ | 10. States of Matter: Gaseous | 10.1 Introduction, 10.8.3 Liquefaction of gases and critical constants, 10.9 Liquid state (Vapour pressure,, and Liquid State Surface Tension and Viscosity only understanding of concepts) Factors affecting vapour pressure, Application, of surface tension., TT | 11. Adsorption and Colloids | 11.1 Introduction ,11.5 Adsorption Isotherm, 11.7.1 Homogeneous Catalysis, 11.7. Heterogeneous catalysis,, 11.73 Inhibitors, 11.8 Adsorption Theory of Heterogeneous Catalysis, 1.9.3 Preparation of colloids, 11.9.4, Purification of colloidal solutions, 11.9.5 e. Electrical Properties, f. Coagulation, | 1.9.6 Methods of coagulation., 12 | 12.Chemical Equilibrium 12.7 Application of equilibrium constant,, 13 | 13.Nuclear Chemistry and 13.2 Classification of nuclides, 13.3.4 Nuclear potential, 13.3.5 Nuclear Binding energy and mass defect, Radioactivity, 14 [14. Basic principle of organic | 14.1 Introduction ,14.6 Theoretical basis of organic reactions,, , chemistry, , , , , , , , , , , , 15 | 15.Hydrocarbon 15.1.3 Industrial preparation of alkanes, Free radical mechanism of halogenation of alkanes, 2. Combustion ,3, Pyrolysis . 4 Reforming a) Industrial source of alkenes . 15.4.2 Structure of Benzene,, 16 | 16.Chemistry in everyday life, , , , Introduction, Traditional knowledge in medicine, Table 16.2 Active ingredients of some medicinal plants,
 Learn better on this topic
Learn better on this topic