Notes of Msc Biotechnology 21-22, 21-22 applied immunology and vi - Study Material
Page 1 :
Applied immunology &virology, Paper No. IX, INDEX, PARTICULARS OF THE EXPERIMENTS PERFORMED, , , , , , , , , , shah hhh pd Md, , , , , , , , , , , , , , , , , , [ Sr. Name of practical Page No. | Signature, No., A) To study purification of immunoglobulin by, “ precipitation \-g, ( 2 To study the isolation of O&H antigen from q~1S, ~ Salmonella typhi, 3 | To study diagnostic assay for Typhoid using \e— 7, WIDAL kit, 4 Phage Isolation from sewage Wen | {, C5) Enzyme Linked Immunosorbent Assay a- 2Q | \, NUYS, 1, , , , , , , , EEE DESSESDSE A TIN SBR Qh Vac Ma Nea ic ei ie seal, , Scanned with CamScanner
Page 2 :
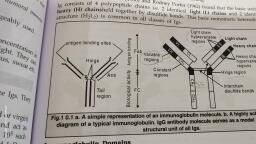
eT nnatam avd viralney, , , , ‘enemas, , Experiment No: 4, , ISOLATION AND PURIFICATION OF IgG, , Aim:, To extract and purify immunoglobulin from given serum sample., , Theory:, , Immunoglobulin functions as antibodies, the antigen binding protein that is present on, B cell membrane and are secreted by plasma cells. Antibodies are glycoprotein. These are, the effector molecules of humoral immunity and constitute of 20-25% of total serum, proteins. Immunoglobulin is divided into water soluble pseudoglobulin and water, , insoluble euglobulin. All immunoglobulin are produced by B lymphocytes in response to, , antigen., , All immunoglobulin shares certain structural features, bind to antigen and participate, , in a limited number of effector functions. Rodney Porter and Gerald Edelman deduced the, , structure of immunoglobulin using proteases (pepsin and papain)., , Immunoglobulin consists of identical heavy and light chains, held together by, disulphide bonds. If immunoglobulins are reduced and acidified, the heavy and light, chains can be separated. Papain digestion of immunoglobulin (IgG) produces two, identical fragments (each with molecular weight 45,000) called F,, fragments because, they have antigen binding activity and the other (molecular weight 50,000) called Fe, fragment because it was found to crystallize during cold storage. Pepsin digestion, , generated a single 100,000-molecular weight fragment composed of two F,-like, fragments, capable of binding to antigen., Antibody molecules have a common structure of four peptide chains. This structure, , consists of two identical light (L) chains (molecular weight 25,000) and two identical, heavy (H) chains (molecular weight 50,000). Each light chain is bound to a heavy chain, , , , 20 -20 [ Mise S17 Sem, Page no:, , Scanned with CamScanner
Page 3 :
re, , , , , , , , rolnow and virology ! pate:, , by disulphide bond. The F, region contains only the carboxyl terminal. Fab consists of, , Portions of both heavy and light chains and lies towards the amino terminals., , There are five classes of immunoglobulin, depending upon the heavy chain. These are, IgG (y), IgA (a), IgM (11), IgD (8) and IgE (¢). The amino terminal parts of the chain,, consisting of 100-1000 amino acids, show great sequence variation and hence called, , variable regions. The remaining part of heavy chain corresponds to constant region., , There are two types of light chains, kappa (x) and lambda (A), these are distinguished, on the basis of amino acid sequence present at carboxyl terminal half of constant region, of the molecule. A single antibody molecule contains only one type of light chain, either, , kappa or lambda light chain, never both., , Immunoglobulin can be purified from any normal serum. AS they are protein, molecules, they get precipitated at specific concentration of salts in solution. Divalent, anions precipitated protein more efficiently than monovalent ions due to effective, , dehydration of protein by salt., , In the present experiment, antibodies were precipitated with ammonium sulphate, as, its salts solutions contains large amount of divalent anions. As the anionic strength of, plasma protein solution increases, the different protein products get precipitated, sequencially. A crude immunoglobulin fraction contains IgG (7), IgA (a), IgM (1), IgD, (8) and IgE (e). At different concentration of ammonium sulphate, different fractions of, immunoglobulins are precipitated. The fraction precipitated by various percentage of, , ammonium sulphate is, , , 25% - Fibrinogen, 35% - IgG, 40% - IgG + IgA + IgM, 50% - a+ B globulin, 10% - Albumin, 20 -20 | MSc. SY. IV Sem. — Dane nn:, , Scanned with CamScanner
Page 4 :
Fino 5, ology and virology 1 Date: ", , These separated immunoglobulins can be confirmed by various techniques such as, , immunodiffusion, immunoelectrophoresis etc., , FUNCTIONS OF IMMUNOGLOBULIN :, IgG:, , ¢ It is the most abundant Ig in serum (80%)., , © It is the only maternal immunoglobulin, which can pass across the placental, , barrier and provide natural passive immunity to new born., , ¢ It binds to microorganisms and enhances their phagocytosis., , e Extracellular killing of target cells coated with IgG is. mediated by antibody, , dependant cell cytotoxicity., It participitates in most immunological reactions such as complement fixation,, , precipitation, neutralization of toxins, etc., IgM:, It provides protection against antigen that has repeating antigenic, , determinants., It is the first immunoglobulin class produced in primary response., , It is 500-1000 times more effective than IgG in opsonization., , It is effective in bacterial action and more against blood invasion by, , microorganisms., , It is present in body secretion and plays a role in local immunity against, respiratory and intestinal pathogens., e Itactivates alternate complement pathway., , Igk :, , e [tis concerned with allergic responses., , Page no: 2, , , , 20 -20 | MSc. S.Y. IV Sem., , Scanned with CamScanner
Page 5 :
* It is protective against pathogen, by mast cell degranulation and release of, , inflammatory mediators., , ° It is effective in helminthic infection., , It is membrane bound immunoglobulin expressed by mature B cells. It is 0.3%, , of total Igs., , Part I- Isolation of IgG, , Requirements :, , 1. 2ml serum sample, , 2. 50% saturated [(NH4)2SOx] ie. 0.1313 gm of [(NH4)2SOq] for 1 ml, serum sample., , Phosphate buffer saline( 20mM, pH 7.2),, , Preparation: 17.4g Dipotassiumhydrogen, , ive), , (K2 HPOs) phosphate & 13.6 g of (KH 2PO4) was added to100 ml of, distalled water pHwas adjected to 7.2 and diluted to 20mM and 0.86, , % of NaCl was added., , Procedure:, 1. 2 ml of serum was taken and to it 0.64 ml phosphate buffer saline was, , added., 2. 1.36 ml of saturated ammonium sulfate solution was added to the above, , mixture., 3. This mixture was incubated at 4°C for 30 — 60 minutes., , 4. — It was centrifuged at 3000 — 4000 rpm for 30 minutes or at 5000 rpm for 10, , minutes., 5. Pellet was dissolved in 1 ml of phosphate buffer saline., , , , Page no: G, , , , 20 -20 ] Se. SY IV Sem., , Scanned with CamScanner
 Learn better on this topic
Learn better on this topic