MCQ Test of 12.Maths...weekly Test, Chemistry Che(chap All) - Study Material
Page 1 :
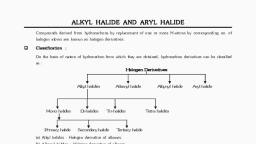
10,, , 12., , VISION SCHOOL OF SCIENCE, NADIAD, , MONTHLY EXAM., Paper ~6, , Std: 12", Sub : Chemistry Chapter ~All, , , , Choose correct answer from the given options. (Each carries 1 Mark), The melting points of ionic solids are ......... ., (A) Normal (B) Very high (C) Abnormal (D) Low, , What is rate of reaction order of the following reaction ?, 43, , Rate = KiX}5(¥>, , 6 5 4, (A) 1 8) 5 Q & (D) 5, Which of the following acids forms three series of salts ?, (A) HPO, (8) H,B0, (©), HPO, (D) HPO,, What is the SI unit of reaction rate ?, (A) mol sec”! (B) mol mS sect (©) mol dm sec (D) mol tir?, FexO contains one Fe(III) for every three Fe(II). What is x ?, w § w ¢ @? wm) 3, Which of the following is the correct order of flocculating for the coagulation of positive ion ?, (A) [Fe(CN)gI*- > $0, > Cr > PO? (B) PO, > [= e(CN)* > S02 > Cr, (©) [Fe(CN),I* » PO? > S0,2> > Cr (D) None of above, , Which of the following should be most volatile ?, , (1) CHjCH,CH,NH, (I) (CH,),N (il) “1 m OV) CCHCH,, , A) aD (B) (IV) ©@ (D) (ap, Which polymer having caprolactum is one of the monomer ?, (A) Nylon 6 (B) Nylon-6,6 (C) Nylon 2 Nylon 6 —_(D) Terylene, , Which of the following compounds is the weakest Bronsted base ?, , “6 NS We AS, , Identify the correct order of oxidizing strength :, , (A) VO$ > Cr,03> > MnO; (B) MnO; > VO$ > Cr,03(©) Cr,03- > MnO; > VO$ (D) MnO; > Cr,03- > VO$, The reaction :, , “mo. ome. 6, ou “, is known as..., (A) Sandmeyer's reaction (B) Balz-schiemann reaction, (C) Birnbaum-Simonini reaction (D) Gatterman reaction, The source of nitrogen in Gabriel synthesis of amines bs ..........., (A) Sodium azide, NaN, (B) Sodium nitrite,, , (C) Potassium cyanide, KCN (D) Potassium phthalimide, C.H,(CO),NR
Page 2 :
When in reaction concentration of reactant is 8 times increases then rate of reactant is doubled than, what is order of reaction ¢, , 1, (A) first ®) 3 (C) 0.5 (D) 0, If dispersed phase and dispersion medium both are solid, this type of sol is called ........., (A) Sol (B) Solid sol (C) Aerosol (D) Emulsion, Which of the following complex ions absorbs the light of minimum wavelength ?, , (A) (CoF,)-* (8) (Co(NH,),)* (©) [Co(CN),)* (D) {Co(H,0),)%*, , What is the ratio of Al, Cu and Na when 3F current is pass through different electrolytic solution of, molten Al,O,; aqueous CuSO, and molten NaCl ?, , , , (A) 3:4:6 (B) 2:1:6 (C) 3:2:1 (D) 2:3:6, The correct decreasing order of basic strength of the following species is .., , H,0, NH,, OH, NH;, , (A) NH3 > OH- > NH, > H,O (B) OH- > NHj > H,O > NH,, , (C) NH, > H,O > NH; > OH” (D) HO > NH, > OH™ > NH3, Which of the following is diamagnetic ?, , (A) Cu?* (B) Zn?* (C) Co?* (D) NF*, Which of the following is isoelectronic pair 7, , (A) ICL, Clo, (B) BrO3s , BrF§ (C) C10, Brk (D) CN", O,, , Which of the following is correct order for boiling points ?, (A) Alcohol > Carboxylic acid > Ether > ketone — (B) Carboxylic acid > Alcohol > Ether > ketone, (C) Ether > Alcohol > Ketone > Carboxylic acid (D) Carboxylic acid > Alcohol > Ketone > Ether, , Fog is an example of ........., , (A) Solid sol (B) Sol (C) Aerosol (D) Gel, Grape SUgAE IS see, (A) Sucrose (B) Glucose (C) Fructose (D) Lactose, , In the extraction of copper from its sulphide ore, the metal is formed by the reduction of Cu,0, with..., , (A) FeS (B) CO (CQ) Cas (D} SO,, , Which of the following complex does not show geometrical isomer ?, , (A) (Cr(Ox),)* (B) (Co(NHy),(NO,),} (C) [Co(NH,),Cl,)* (D) [Fe(NH,),(CN),)At which temperature, ceramic matters are known as super conductor ?, , (A) OK (8) 200 K (C) 150 K (D) 15 K, , Which of the following Fisher projection formula is identical to D-glyceraldehyde ?, , CH,OH, , om CH,OH CHO, A) Ho} co qe tf-ou () 1} eon, x CHO OH, , H, , CH, - CH, ~ NH, + CHCl, + 3KOH —4» X + 3KCl + 3H,0. What is X ?, , (A) Ethyl isocyanide (B) Ethyl cyanide (C) Propane nitrile (D) Ethanamide, What is the vapour pressure of the solution prepared by dissolving 0.5 gram substance (Molecular, weight 65) in 100 m! CCl, ? The vapour pressure of CCl, is 143 mm. density of CCl, is 1.58., , (A) 143.99 mm (B) 94.39 mm (C) 141.97 mm (D) 199.34 mm, When initial concentration of the reactant is doubled, the half-life period of a zero order reaction..., (A) remains unchanged. (B) is halved. (C) is tripled. (D) ts doubled., , Which of the following ores are concentrated by froth floation ?, (A) Haematite (8) Galena (C) Copper pyrites (D) (B) and (C), , Hit
Page 3 :
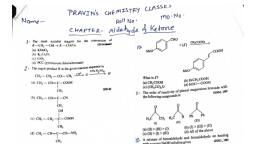
3., , 32., , 3s., , 37., , 42., , Which of the following is correct for P, molecule of white phosphorus ?, , (A) It has 6 lone pairs of electrons. (B) It has six P-P single bonds., , (C) It has four lone pairs of electrons. (D) (B) and (C), , If ACICH,COONa = 224 Ohm=! cm? gm equ’, ANaCl = 38.2 Ohm=! cm? gm equ! and AHCI = 203, Ohm” cm? gm equ, then what is the value of XCICH,COOH ?, , (A) 288.5 Ohm=! cm? gm equé! (B) 289.5 Ohm! cm? gm equ!, , (C) 388.5 Ohm cm? gm equ (D) 59.5 Ohm! cm? gm equ"!, , Which of the following statements are correct ?, , (A) S-S bond is present in H,S,0,., , (B) In peroxosulphuric acid (H,SO,) sulphur is in (+6) oxidation state., , (C) Iron powder along with Al,O, and K,O is used as a catalyst in the preparation of NH, by Haber's, , process., (D) (A) and (B), Which linkage is present in disaccharide ?, (A) Amide (B) Glycosidic {C) Phosphodiester (D) Carboxylic, , The coagulating power of electrolytes having fons Na‘, Al’* and Ba** for arsenic sulphide sol, increases in the order ;, , (A) Al* < Ba?* < Na* — (B) Na* < Ba®* < AP (C) Ba®* Nat < Al* = (D) AP* < Nat < Batt, Which complex ion of the following possess 2.82 BM magnetic moment ?, , (A) Ni(CO), (Dy ENICAg)?> (C) Ni(PPhy) , (D) [Ni(CN),)”, Which of the trivalent tons is colourless ?, (A) Gd (B) Pm3* (©) Im* (D) B+, , What is strong basicity order ?, (A) NH > Clg > (CyHgN>(CaHt)y—NH> 4B) (C3P)gNH > (Cly)y N> Gyll,NH > NH,, , (©) (Collg)gNH> Clg NIly > (CyMl)gN >My €D)(C4ltsN> (Colly)y NHL > CallgNtt, > NI, The major product in the reaction is..., , jo, ino HI, (CHOH), <p ed, , nm, , (A) Heptanoic acid (B) n-hexane (C) Hexanedioc acid (D) Saccharic acid, Which of the following cannot be prepared by Sandmeyer's reaction ?, , (A) lodobenzene ({B) Fluorobenzene {C) Chlorobenzene (D) (A) and (B), Which of the following is not a natural polymer ?, , (A) Starch (B) Cellulose (C) Nylon (D) Protein, , Which complex gives 18.8 gms of precipitates when 0.1 M aqueous solution of AgNO, is added to, its 1 L 0.1M aqueous solution ? (Mole. mass of AgBr = 188 gm mol!), , (A) Penta ammine bromido cobalt (II) bromide — (B) Tetra ammine dibromido cobalt(II!) bromide, (C) Potassium diamminete trabromido cobaltate(IIl) (D) Tri ammine tri bromido cobalt (III), , Which of the following complex sp*d? type hybridization ?, (A) [Fe(NHy)6)°* (B) [Re(Cl),)* (C) [Fe(CN)_)*- (D) [Fe(CN),)*, Sodium metal crystallizes in a body-centred cubic lattice with a unit cell edge of 4.29 A. The radius, , of sodium atom is approximately .......... ., (A) 5.72 A (B) 0.93 A (C) 1.86 A (D) 3.22 A, , The molal depreesion constant of water is 1.86°C/m. Then find out the freezing point of 0.1 m NaCl, solution., , (A) -1.86° C (B) 0.372° C {C) -0,372° C (D) -0.186° C
Page 4 :
The half life period for a first order reaction is, , (A) proportional to concentration., , (B) independent of concentration., , (C) inversely proportional to concentration., , (D) inversely proportional to the square of the concentration., , 47. Which is a best substance for coagulation of gold sol ?, (A) KNO, (B) Ky|Fe(CN),) (C) MgCl, (D) K,PO,, 48. How many unit cells are present in a cube shaped ideal crystal of NaCl of mass 1.00 g ?, [Atomic masses : Na = 23, Cl = 35.5), (A) 2.57 x 1022 (B) 5.14 « 1022 (C) 1.28 x 10% (D) 1.71 « 1072, 49, Common impurities present in bauxite are....., (A) CuO (B) ZnO, (C) Fe,0, & SiO, (D) None of the above, 50, Reaction of anisole with conc, HNO, and cone, H,SO, will give..., (A) phenol (B) ortho nitro anisole (C) nitro benzene (D) ©, p-nitro anisole, PART -B, SECTION =A, * Answer the following questions; [Each carries 2 mark] [ANY 8] (u, , Peravayne, , Difference between Ideal and Non-Ideal Solution., Explain about Excess Metal defect., , Explain Pseudo first order reaction., , Hardly and Schulze rules for coagulation, , Difference between Physical & Chemical Adsorption., Concentration of metal by Leaching., , Allotropes of Phosphorus., , Oxo Acids of Sulphur., , Hume & Rothery Rule,, , . Write a short note on Sandmeyer's reaction?, . Hell — volhardzelinsky reaction., . Explain reaction Hoffmann reaction, , SECTION ~B, Answer of the following Question: (Each carries 3 mark] [ANY 6] (u, , . Explain (i) Schottky defect (ii) Frenkel defect, , . Explain Raoult’s Law & limitation for non-volatile solute., , . Freundlich adsorption Isotherm., , . Extraction & uses of Iron (Fe)[Blast furnace), , . Preparation, physical & chemical properties of Ch,, , . Werner's Theory and its point of COT., , . Write short note on Reimer-Tiemann and Kolbe-smith reaction?, , . Type of Rubber? (natural / vulcanized), , . preparation and uses of addition polymer; a.HDP ethane b. polystyrene c.SBR, SECTION ~C, , newer tho tiliening questions ns divested bn the question. [Roch enevies 4 mest] ANY @ {u, , 22, Calculate of atom in unit cell, Radius and Packing Efficiency in BCC,, , 23. Explain Nerst Equation for two cell potential, equilibrium and concentration cell., 24, Extraction & uses of Aluminium (Al) [Baeyer's process and Hall-Heroult process}, 25, Preparation, Properties and Uses of (i) KaCr)0y (ii) KMn0,., , 26. Derive rate constant and half life time for Zeroth order reaction., , 27. Aldol, cross aldo! condensation and canizarro reaction
 Learn better on this topic
Learn better on this topic