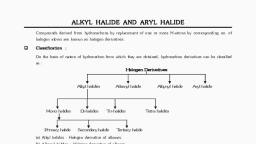
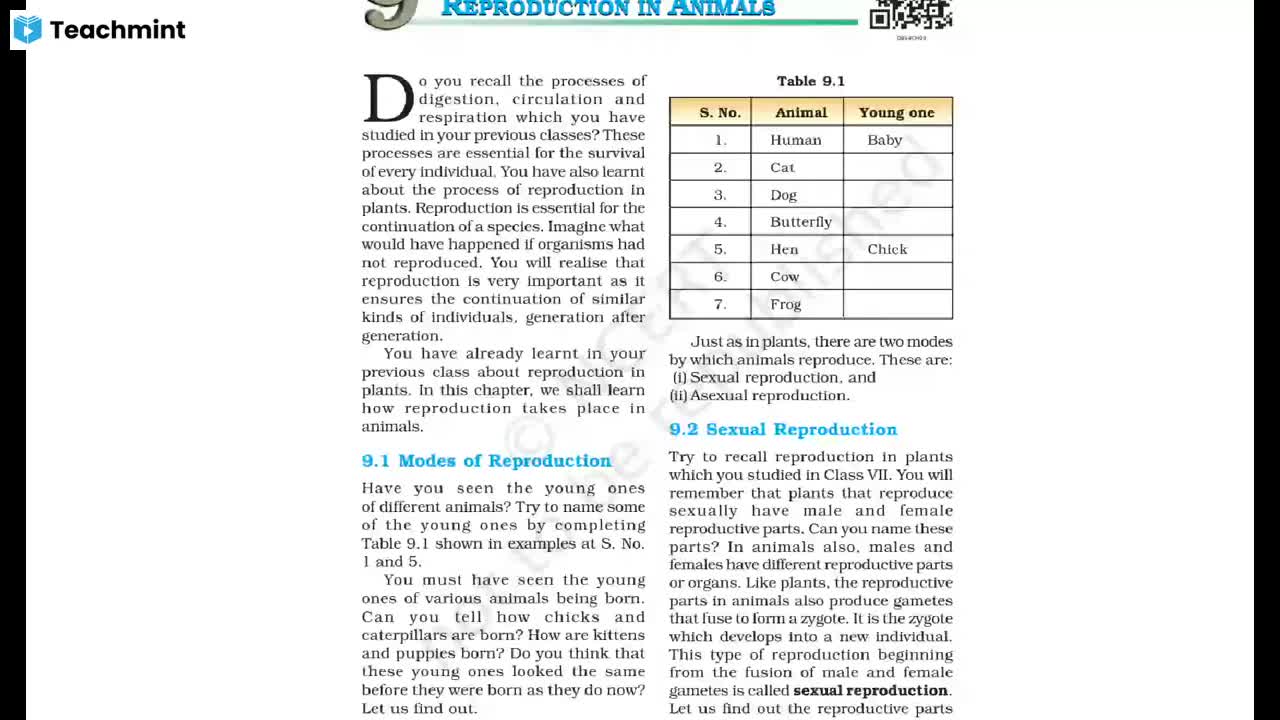
MCQ Test of 12th CHEMISTRY, Chemistry Halogen Derivative - Study Material
Page 1 :
Objective Questions, 05. Which of the following is an, vic dihalide?, example of, (NCERT Exemplar), (b)1, 2-dichloroethane, Multiple Choice Questions, (a) Dichloromethane, (c)Ethylidene chloride (d)Allyl chloride, 06. In which of the following molecules, carbon atom marked with asterisk ()i, 01. Which is the correct IUPAC name for, CH, — СH—СН, — Br ?, (NCERT Exemplar), C,H,, (a)l-bromo-2-ethylpropane, (b)1-bromo-2-ethyl-2-methylethane, (c)1-bromo-2-methylbutane, (d)2-methyl-1-bromobutane, asymmetric?, D, H., II., I., CI, 02. Compounds in which the halogen atom, is bonded to a sp²-hybridised carbon, atom of a C-C double bond is known as, (b) benzylic halide, (d) vinylic halide, Br, Br, H., IV., CH3, (a) allylic halide, (c)aryl halide, 03. Which of the following is benzyl, halogen compound?, (a) Br-CH,CH(CH,)CH,CH,, (b) (CH,1,CCH,CH(BriC,H,, (c) (CH,), CHCH(CI)CH,, (d) None of the above, 04. Match the structures of compounds, given in Column I with the classes of, compounds given in Column II., III., CH3, OH, CHs, C,H5, (b)l, Il and II, (d)I, IIl and IV, (a)l, II, IIl and VI, (c) II, III and IV, 07. Possible number of isomers of C,H,Cl,, compound are, la)2, (c)6, (b)4, (d) 8, 08. Match the structures given in Column I, with the names in Column II., Column I, Column II, Column I, Column II, A. CH,-CH-CH,, 1. Aryl halide, А., Br, 1. 4-bromopent-2-ene, B. CH,=CH-CH,-X 2. Alkyl halide, В., 2. 4-bromo-3-, 3. Vinyl halide, Br, methylpent-2-ene, С., 3. 1-bromo-2-methylbut, -2-ene, D. CH,=CH-X, 4. Allyl halide, Br, D., Br, Codes, 4. 1-bromo-2-methyl-, pent-2-ene, (NCERT Exemplar), ABCD, (a) 2 4 1 3, (b) 3 2 1 4, Ie) 43 21, РОСО 4 2, Codes, А В С D, (a) 1 2 3 4, (c) 2 3 4, (NCERT Exemplar), ABCD, (b) 4 3 2 1, (d) 4 3 1 2, SHOT ON POCO F1, 1.
Page 2 :
0g. Which of the following structures is, enantiomeric with the molecule (4), given below?, 14. Select the by product formed in the, reaction,, H, ROH+ PCI,, Here, A is, → RCI +'A', CH3, (a) POCI, + HCI, (b) H,PO, +No other by-product, (cl H,PO, + HCI, (d) POCI, +No other by-product, HSC, Br, (4), H., CH, CaHs, 15. The order of reactivity of following, alcohols with halogen acids is, A. CH,CH,-CH,-OH, B. CH,CH,-CH-OH, (a), (b), HC, .*******.., Br, Br, C2HS, H., Br, Br, ost, (d), H;C2, (c), H3C, C2HS, CH,, CHa, CH,, 10. Molecules whose mirror image is, non-superimposable over them are, known as chiral. Which of the following, molecules is chiral in nature?, C. CH,CH,-, OH, C-, CH,, (NCERT Exemplar), (a)(A)>(B)>(C), (c)(B)> (A)>(C), (b)(C)>(B)>(A), (d)(A)>(C)>(B), (a) 2-bromobutane, (c)2-bromopropane, (NCERT Exemplar), (b)1-bromobutane, (d)2-bromopropan-2-ol, 16. The alkane that gives only one, monochloro product on chlorination with, Cl, in presence of diffused sunlight is, (a) 2.2-dimethylbutane (b) neo-pentane, (c) n-pentane, 11. What is the name of the following, reaction?, Nal, CH,CH,CH,Br, Dry acetone, (d) isopentane, 17. The reagent used in Gattermann's, reaction is, CH,CH¸CH,I, (a) Sandmeyer reaction, (b) Gattermann reaction, (c) Finkelstein reaction, (d) Swarts reaction, ......, (b) CuBr/HCI, (d) KI, (a) Cu/HBr, (c)CuCl,/HCI, react with Cl, in presence of, light and then react with Na metal in, dry ether, final product is, 18. The product formed on the reaction of, Br, with silver benzoate in the presence, of CCI, and heat, is, (a)C,H,Br, (c)C,H,Br, 12. If, ......, (b)C,H,Br,, (d)C,H,Br,, Br, (a), (b), 19. Chlorobenzene is formed by reaction of, chlorine with benzene in the presence, of AICI,. Which of the following, species attacks the benzene ring in this, reaction?, (c) Z, (d), 13. Alkyl halides are prepared from, POCOS by treating with, SHOT ON POCO F1, tc) H, So, + KI, (NCERT Exemplar), (a)Cr, (b)CI, (b) Red P+ Br,, (d) Both (a)and (b), (c) AICI,, (d)(AICI,1
Page 3 :
62, 24. Carbon-halogen bond is polarised because, (a) halogen atom is less electronegative, (b) halogen atom is more electronegative than, carbon atom, 20. Chlorination of toluene in the, of light and heat followed by treatment, with aq. NaOH gives, (alo-cresol, (blp-cresol, (c)mixture of o-cresol and p-cresol, (d)benzoic acid, presence, (c) carbon bears negative charge while halogen, bears positive charge, (d) None of the above, 21. Arrange the following compounds in, increasing order of their boiling points., CH,, I., 25. Which of the following alkyl halides has, maximum density?, (a)C,H (b)C,H,!, (c)CH,Br (d)CHJ, 26. Arrange the following compounds in the, increasing order of their densities., CH-CH, Br, CH,, II. CH,CH,CH,CH,Br, CH,, I., II., III. H,C-C-CH,, Cl, Br, Br, (NCERT Exemplar), III., IV., (b)l < || < II, (d)III < II <I, (a)ll<l< II, (c) II <l< ||, Cl, Cl, (NCERT Exemplar), (b)l < III < IV < II, (d) || < IV < III <l, 22. Which is the correct increasing order of, boiling points of the following, compounds?, 1-iodobutane, 1-bromobutane,, 1-chlorobutane, Butane, (a) Butane <1-chlorobutane < l-bromobutane <, 1-iodobutane, (a)l< || < |II < IV, (c)IV < III < || <I, 27. Arrange the following compounds in the, increasing order of their melting points., CI, (b) 1-iodobutane <1-bromobutane <, 1-chlorobutane < Butane, (c) Butane <1-iodobutane <1-bromobutane <, 1-chlorobutane, (d) Butane <1-chlorobutane < 1-iodobutane <, 1-bromobutane, II, III, (a) I> II> II, (c) III >1211, (b) III >1 = |, (d) III >I> I|, 23. Arrange the following compounds in, order of increasing dipole moment:, Toluene (I), m-dichlorobenzene (II),, o-dichlorobenzene (III),, p-dichlorobenzene (IV)., (a)l<IV < |l < II, (b)IV<l<ll < II, (c) IV<I< II< ||, POCO kl<.l, 28. Which of the following has the highest, melting point but least solubility in a, given solvent?, (a)o-dichlorobenzene (b)p-dichlorobenzene, (c)m-dichlorobenzene (d) Chlorobenzene, 29. Arrange the following compounds in, increasing order of their density., I. CH,CI, II. CHCI, III. CCI,, (a) II < || <I (b) II<l< |I (c) II < = || (d) I<||<|, SHOT ON POCO F1
Page 4 :
A The main product of reaction, CH, CHCH,CH,, 1., Br, 35. Consider the following reactions:, ÇH5, H, Ale. KOH, ?, (II) xH, CH, (1), HC, CH3, (a)Propene, (c) Butane, (b) Butene, B, (d) Butyene-1, A+B, 41. Which of the following alkyl halides, will undergo S,I reaction most readily?, (NCERT Exemplar), (b)(CH,),C-CI, (d)(CH,),C-I, Name the terms that occur in I, II, III, equations. Choose the correct option., II, III, (a)(CH,),C-F, (c(CH,),C-Br, (a) Retention of, configuration configuration, (b) Retention of, configuration, Inversion of, Racemisation, 32. Which of the following alkyl halides is, hydrolysed by S, 2 mechanism?, (a)C,H,CH,Br, (c)CH, = CHCH,Br, Racemisation Inversion of, configuration, (c) Racemisation Retention of, Inversion of, (b)CH,Br, (d)(CH,),CBr, configuration configuration, Retention of, configuration configuration, (d) Racemisation Inversion of, 33. Which of the following compounds will, give racemic mixture on nucleophilic, substitution by OH¯ ion?, 36. Match the items of Column I and, Colum II., I. CH,-CH-Br, Column I, Column II, C,H,, A. Syl reaction, 1. vie-dibromides, Br, B. Chemicals in fire, extinguisher, 2. gem-dihalides, II. CH, — С— CH,, C. Bromination of, alkenes, 3. Racemisation, CH,, D. Alkylidene halides 4. Saytzeff rule, III. CH,-CH-CH,Br, E. Elimination of HX 5. Chlorobromo, (NCERT Exemplar), from alkyIhalide, carbons, C,H,, (NCERT Exemplar), (a) I, (c)Il and III, (b) I, Il and II, (d)l and II, Codes, ABCDE, ABCDE, (a) 3 5 1 2 4, (c) 2 5 3 4 2 (d) 1 2 3 4 5, (b) 5 1 342, 34. Cyanide acts as an ambident, nucleophile. From which of the, following aspects it acts as a stronger, aqueous, la) It has same strength from both the ends, (b)It depends on the nature of alkyl halide, (c) It acts as a stronger nucleophile from N-end, as a stronger nucleophile from C-end, 37. Reaction of C H,CH,Br with aqueous, sodium hydroxide follows, (a) S,1mechanism, (b) S,2 mechanism, (c) Any of the above two depending upon the, temperature of reaction, (d) Saytzeff rule, nucleophile in, medium?, ..*...**.., (NCERT Exemplar), POCO, SHOT ON POCO F1
 Learn better on this topic
Learn better on this topic