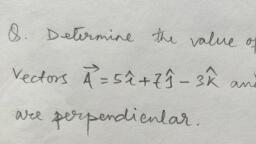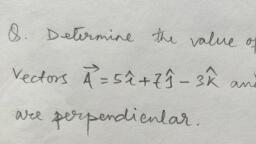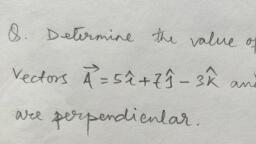MCQ Test of Medical, Physics Heat, Thermodynamics & KT of Gas - Study Material
Page 1 :
1, , KK’s TUITION CENTRE, , MEDICAL AND ENGINEERING PHYSICS, FORTNIGHTLY TEST-7, (Heat, Thermodynamics & Kinetic theory of gas), , Attempt all questions and each question carries 4marks., Each wrong answer carries (-1) mark., FM: 120, , Time : 40 min, , 0.1m a is mixed with 0.3 m’ of water at 60°C. The final temperature of the mixture is, , a) 63°C., , b) we Ans°| |, , ©) 60°C., , d) 75°C., A lead bullet strikes against a stee! plate with a velocity 200 ms". If the impact is perfectly inelastic and the heat, produced ts equally shared between the bullet and the target, then the rise in temperature of the bullet 1s (specific, heat capacity of lead = 125 J kg’ K"), , a) 60°C., , b) 80°C Ans:[ J, , ©) 160°C., , d) 40°C, The rods of equal length and diameter have thermal conductivities 2 and 3 units If they are joined in series, the, thermal conductivity of the combination will be, , a) lunit, , b) Sunits Ans: [ ], , c) bunits, , d) 24units, A black body at 1227°C emits radiations with maximum intensity at a wavelength of 5000 A. If the temperature, of the body is increased by 1000°C, the maximum intensity will be observed at, , a) SOOOA Ans:[ ], , b) 4000 A, , ©) 60004, , d) 30004, One end of a thermally insulated rod is kept at a temperature T, and the other at T,. The rod is composed of two, sections of lengths /, and /, and thermal conductivities k, and k, respectively, The temperature at the interface of, the two sections 1s, , a) Hehehe, kth,, , kl Ty shoals To, >) Key dy t hel Ans-[ J, , ¢) taeTitkliTs, Wylyohaly, , B17, 6k AT,, o eae, A body cools from 80°C to 64°C in 5 min and same body cools from 80"C to 52°C in 10 min, What is the, temperature of the surroundings?, , (a) 4°C, , (b) 28°C Ans'[ J, , (c) 22°C, , (d) 25°C, An ideal monoatomic gas at 27°C 1s compressed adiabatically to 8/27 times of its present volume. The increase in, temperature of the gas is, , (a) 179°C., , (b) 375°C. Ans-[ ], , (c) 402°C
Page 2 :
10, , 12, , 14, , 15, , 16, , (d) 475°C., A Carnot engine whose sink is at 300K has an efficiency of 40%. By how much should the temperature of the, source be increased so as to increase its efficiency by 50% of original efficiency?, , (a) 275K., , (b) 325K. Ans:[ ], , (c) 250K ., , (d) 380K., When a system ts taken from state 1 to state f along path taf, it ts found that Q=50 cal and W=20 cal, Along the, path ibf, if Q=39 cal, then the value of W along the path ibf is, , (a) eal., , (b) 14 cal. a f, , (c) 19 cal tt Ans:| |, , (d) 26 cal., , , , , , , , , , ‘The internal energy change in a system that has absorbed 2 kcal of heat and done 500 J of work as, (a) 400), (b) 64003, (c) 79003 Ans-[ ], (d) 8900), , Starting with the same initial conditions, an ideal gas expands from volume V; to volume V> in three different, ways. The work done by the gas is W, in isothermal process, W, in adiabatic process and W, in isobaric process., Then, , (a) W.> WW;, , (b) W3> Wy W; Ans'[ J, , (c) Wa> W3> W2, , (d) Wy We W,, When 2 moles of oxygen is heated from 0°C to 10°C at constant volume, its internal energy changes by 420 J, What is the molar specific heat of oxygen at constant volume ?, , (a) $.75JK"'mol™, , (b) 10.5JK"'mor! Ans:[ |, , (c) 21 JK"mot', , (@) 42 JK"'mol', A monoatomic gas at pressure P, and volume V, is compressed adiabatically to 1/8* its original volume. What is, the final pressure of the gas’?, , (a) Py, , (b) 16P,, , (c) 64P, Ans.[ ], , (d) 327,, A diatomic ideal gas is compressed adiabatically to 1/32 of its initial volume. If the initial temperature of the gas, ts T{in Kelvin) and the final temperature is aT, the value of ais, , (a), , (b) 2 Ans:[ ], , (c) 4, , (d) 6, , When the temperature of a rod increases from t to t-At, its moment of mertia increases trom I to I+AL. If a is the, coefficient of linear expansion of the rod, then value of ALT is, , a) 2adt, , b) aat Ans:[ |, , c) a@dv2, , d) Av2, , A refrigerator with coefficient of performance 1/3 releases 200 J of heat to a hot reservoir, Then the work done, on the working substance is, , (a) 200/3 J., , (b) 2003. Ans:[ ], , (c) 10/3 J, , (d) 150)
Page 3 :
17., , 18., , 19., , 20., , 2, , 23., , 24, , 25., , 26, , 3, , 420 J of energy supplied to 10 g of water will raise its temperature by nearly [specific heat of water = 4.2x10*, Jkg"K"'|, , a VC, , b 42°C, , c. WC Ans:[ ], , d 42°C, A two litre glass flask contains some mercury. It ts found that at all temperatures the volume of the air inside the, flask remains the same. The volume of Hg inside the flask is [@ for glass= 9x10 °C"; y of Hg = 18x10" °C"}, , (a) 1500 ce., , (b) 150 cc. Ans:{[ ], , (c) 00 ce., , (d) 300 ce., If on heating a liquid through 80°C, the mass expelled ts 1/100" of mass still remaining, the coefficient, expansion of the liquid ts, , (a) 125x104 °C"., , (b) 125x104 °C"., , (ec) 125x107 °C". Ans:[ |, , (d) 125x107 °C", ‘The volume of metal sphere increases by 0.15% when its temperature ts raised by 24° C. The coefficient of linear, expansion of metal is, , fa) 2.5x10° °C", , (b) 2.0x10° "Cc" Ans:[ ], , (c) LSx10% °C", , (@) 12108 °c", Certain amount of heat is given to 100 g of cu to increase its temperature by 21°C. If the same amount of heat is, given to 50g of water, then the rise in its temperature is ( S_.= 400Jkg'K" and Syye= 4.2x10" Jke'K''], , (a) 4°C., , (b) $.25°C Ans:| |, , (c) 8°C, , (d) 6°C, , _ 0.1m? water at 80°C is mixed with 0.3m’ of water at 60°C. the final temperature of the mixture is, , (a) 63°C., (b) 70°C., (c) 60°C a, (d) 73°C, Hailstorm at 0°C falls from a height of | km on an insulating surface converting whole of its K-E. into heat. What, part of it melts?, (a) 1/33., (b) 18., (c) 3/4. Ans-[ |, (d) All of it will melt, A gas is heated at constant pressure, The fraction of heat supplied use for external work ts, (a) 1/y., (b) L-Ly., (c) y-1 Ans:[ ], (4) Ll y?, At 10°C the value of density of a fixed mass of an ideal gas divided by its pressure is x. At 110°C this ratio is, fa) x., (b) 383/283., (c) WIL. Ans.[ ], (d) 283x/383., The equation of state of Sg of oxygen at a pressure P and temperature T, when occupying volume V will be, , (a) PV-SRT, (b) PV= SRT.
Page 4 :
27, , 28., , 29, , 30., , +, , (c) PV==RT Ans:[ |, , (4) PV=SRT, At constant pressure, the ratio of increase in volume of an ideal gas per degree rise in Kelvin temperature to its, original volume is [T» absolute temperature of gas}, , fT, , (b) T. Ans:[ |, , (ce) VT., , i) vr, The temperature at which the velocity of oxygen will be half that of hydrogen at NTP is, , (a) 1092°C, , (b) 1492°C Ans:[ ], , (ce) 273°C, , (d) 819°C, ‘Two perfect gases at absolute temperature T; and T; are mixed There ts no loss of energy If the number of, molecules tn the gases are n, and n; respectively, then the temperature of mixture of masses of molecules is,, , (a), , mint, (b) as Ans:[, , ntent, ©) ome, , @ [22, nh, The molar specific heat at constant pressure of an ideal gas is 7R/2. The ratio of specific heat at constant pressure, to that at constant volume is, a WS, b 87, ce 57 Ans.[, d 97, , treeeerenee
 Learn better on this topic
Learn better on this topic